Nathalia José Gamarra Martínez2, María Claudia Niño Mejías1, Darwin Cohen1, Andrés Obando Rondón1, Daniel Santiago Benitez1, Fernando Raffán1
Recibido: 2025-09-23
Aceptado: 2025-09-23
©2026 El(los) Autor(es) – Esta publicación es Órgano oficial de la Sociedad de Anestesiología de Chile
Revista Chilena de Anestesia Vol. 55 Núm. 2 |https://doi.org/10.25237/revchilanestv55n2-07
PDF
Alteraciones en la coagulación en pacientes con neoplasias cerebrales. Estudio descriptivo observacional prospectivo de cohorte
Abstract
This descriptive observational study included 60 subjects with brain neoplasms scheduled for surgical resection. They were consecutively sampled to evaluate the global function of hemostasis using static and dynamic tests to establish the relationship between the diagnosed coagulation profiles and intraoperative and postoperative outcomes. To define the relationship between variables, Pearson and/or Spearman correlation analysis, t-test or biserial point correlation test, chi-square test, Cramer’s V test, or Mann-Whitney U test were applied according to the type of variables to be correlated. From the present study, it can be concluded that although thromboelastography (TEG) as a dynamic coagulation test has been shown to be an effective method for assessing coagulation status, no relationship was observed between coagulation patterns obtained from thromboelastography and perioperative outcomes such as hospital stay, intensive care unit stay, and mortality. However, TEG was more efficient in identifying altered states of the coagulation profile compared to what was observed with static coagulation tests, suggesting that the performance of hematologic screening in patients taken to brain tumor resection could be optimized with viscoelastic testing should this resource be available.
Resumen
This descriptive observational study included 60 subjects with brain neoplasms scheduled for surgical resection. They were consecutively sampled to evaluate the global function of hemostasis using static and dynamic tests to establish the relationship between the diagnosed coagulation profiles and intraoperative and postoperative outcomes. To define the relationship between variables, Pearson and/or Spearman correlation analysis, t-test or biserial point correlation test, chi-square test, Cramer’s V test, or Mann-Whitney U test were applied according to the type of variables to be correlated. From the present study, it can be concluded that although thromboelastography (TEG) as a dynamic coagulation test has been shown to be an effective method for assessing coagulation status, no relationship was observed between coagulation patterns obtained from thromboelastography and perioperative outcomes such as hospital stay, intensive care unit stay, and mortality. However, TEG was more efficient in identifying altered states of the coagulation profile compared to what was observed with static coagulation tests, suggesting that the performance of hematologic screening in patients taken to brain tumor resection could be optimized with viscoelastic testing should this resource be available.
Introduction
Cancer is the leading cause of death worldwide, and according to the World Health Organization (WHO), in 2020, almost 10 million deaths were attributed to this disease. In Colombia, this disease is considered a public health problem because it generates an economic burden represented by the lack of productivity, the costs involved in treatment, the impact on the quality of life of survivors, and the years lost as a result of premature deaths[1],[2]. According to Globocan data in Colombia in the year 2020, central nervous system cancer occupies the 16th place corresponding to 1.7% of all new cancer cases reported; however, it occupies the 11th place in terms of mortality with 1,636 cases of the total number of cases in Colombia[3]. Brain tumors of primary origin, whether benign or malignant, are mainly classified according to histopathological criteria updated by the WHO as astrocytoma, ependymoma, meningioma, and embryonal brain tumors that are usually present in the pediatric population, such as medulloblastoma and supratentorial primitive neuroectodermal tumors[4]. Chronic activation of the coagulation system by producing intratumoral vasoocclusive thrombosis, contributing to hypoxia, necrosis, and angiogenesis, has been described in several cases of brain neoplasms. An example is glioblastoma multiforme (grade IV glioma), which has a highly infiltrative growth pattern and is associated with spontaneous or iatrogenic bleeding and the emission of procoagulant agents[4].
Surgical resection is the mainstay for the treatment of neoplasms of the central nervous system; however, these procedures are often complicated by disorders of the coagulation system, which can be secondary to intratumoral hemorrhages, perioperative thromboembolic complications, or coagulopathies, whether congenital or induced by chronic use of drugs such as antiplatelet agents, anticoagulants, or certain naturopathic drugs[5]. Thrombosis is a serious postsurgical complication, especially in patients with brain tumors, such as gliomas or malignant tumors, who present a high risk of thromboembolic complications. The incidence of deep vein thrombosis in these patients varies between 10.3% and 21.3%, and increases in cases of malignant gliomas, where it can reach between 16.1% and 33.3%. In addition, an incidence of pulmonary embolism between 0.5% and 1.5% has been reported, as well as perioperative cerebral ischemia ranging from 27.7% to 50%[6] The main risk factors include advanced age, prolonged surgery time, pre-existing coagulation disorders, and intraoperative bleeding[6]. These coagulation system complications are associated with significant mortality and decreased survival[7]. Therefore, early identification of the population with risk factors, history, and clinical symptoms suggestive of underlying coagulopathies is crucial, as well as early diagnosis through a complete evaluation of the hemostatic profile, which will allow adequate guidance for therapeutic decisions[8].
Viscoelastic coagulation tests are nowadays categorized as dynamic tools implemented to better understand the causes of bleeding, to evaluate hypocoagulable and hypercoagulable states, to guide transfusion therapies and to predict the risk of bleeding during anesthetic or surgical procedures[9],[10], reasons that fill us with reasons to want to implement these tests in patients with brain tumors who are scheduled for surgical resection. The aim of the present study was to identify preoperative coagulation disorders using dynamic coagulation tests and their potential association with the development of thromboembolic or hemorrhagic complications in the perioperative period, and to compare them with static coagulation tests. Additionally, with the development of the present study, we characterized the types of brain neoplasms experienced by patients attending a fourth-level Colombian hospital.
Methods
Study design
This descriptive observational prospective cohort study aimed to evaluate the coagulation profiles of adult patients with brain neoplasms scheduled for surgical resection at a fourth-level hospital in Colombia. The protocol was approved by the Research Ethics Committee of Fundación Santa Fe de Bogotá Hospital in accordance with established ethical principles and current regulations. The sampling was consecutive, selecting patients who met all the inclusion criteria, which consisted of men and women over 18 years of age with benign or malignant neoplastic brain pathology scheduled for surgical resection between March 2023 and March 2024. Exclusion criteria were history of coagulation disorders or hematologic diseases, hepatic or renal diseases, use of homeopathic, anticoagulant, or antiplatelet drugs, and pregnancy.
Sample collection was carried out by members of the research team, who conducted an exhaustive review of the clinical histories of the selected patients. The information collected was recorded electronically on the RedCap platform, ensuring confidentiality of the data and compliance with the recommendations of the institution’s Corporate Research Ethics Committee. Regarding the anesthetic intervention, all patients were managed by the treating anesthesiologist under a standardized technique of total intravenous anesthesia, which included the use of Propofol, Remifentanil, Lidocaine, Dexmedetomidine, as well as basic ASA monitoring. Intraoperative monitoring included arterial line, central venous pressure, and Bispectral Index (BIS) according to the clinical guidelines established for this type of procedure.
Outcomes
The primary outcomes evaluated were estimated intraoperative bleeding, blood product transfusion volume, reoperation for postoperative bleeding, and thromboembolic complications as additional clinical diagnoses recorded in the clinical history (deep vein thrombosis, pulmonary thromboembolism, and acute myocardial infarction). Secondary outcomes were 30-day mortality, days of intensive care unit stay, and hospitalization.
Statistical analysis
Descriptive analysis of the data was performed using measures of central tendency according to the distribution of the variables. Means and standard deviations were used for variables with normal distribution (Figure 1), and medians with interquartile ranges (IQR) were used for variables that were not normally distributed. Normality was assessed using the Shapiro-Wilk test. The differences between quantitative variables were assessed using the Mann-Whitney U test and unpaired t-test according to their distribution. The Chi-square test was used to evaluate the independence of the qualitative variables. Pearson and Spearman correlation coefficients were applied to determine the association between two variables, according to their distribution and nature. All statistical analyses were performed using STATA SE v. 17.0.
Laboratory tests
Patients were required to have a blood count and coagulation time at the time of the pre-anesthetic assessment. All patients had a sample of 2 mL of whole blood taken at the time of cannulation of the arterial line for thromboelastography (TEG) and fibrinogen after anesthetic induction and always before starting surgery. Patients were classified according to the coagulability index of the thromboelastogram (as recommended by the manufacturer of the TEG 5000 equipment) into a normal coagulation profile (CI -3 to 3), hypocoagulable profile (CI < -3), and hypercoagulable profile (CI > 3). Regarding static coagulation tests, fibrinogen level was considered normal in the range established by the institution’s laboratory at 200-400 mg/dL. Platelets were considered to have normal parameters, between 150,000 and 400,000. Coagulation times were considered abnormal when they were 1.5 times the daily institutional laboratory control value or an INR > 1.5. Intraoperative bleeding, the amount in milliliters of packed red blood cells administered (300 mL per unit), fresh frozen plasma (150 mL per unit), apheresis platelets (200 mL per unit) and cryoprecipitates (15 mL per unit) were also recorded.
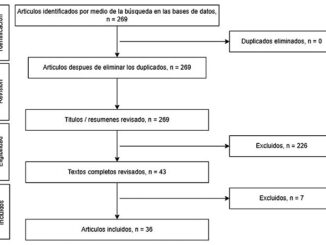
Figure 1. Flowchart of sample distribution.
Results
Sixty patients were enrolled between March 2023 and March 2024, of which 3 (5%) were excluded because they did not have all the static coagulation tests, leaving a total of 57 patients. The demographic and clinical characteristics of patients are shown in Table 1. Demographic data: Patient ages ranged from 19 to 80 years, with a mean age of 48.63 years and standard deviation of 15.28. The included sample is 57.89% female and 42.11% male, with body mass index with a median of 24.82 (RIQ 22.85 – 26.13) Histopathology: 50.88% were benign brain tumors and 49.12% were malignant. The most frequent lesions were meningiomas (33.33%), followed by pituitary tumors (29.8%), glioblastomas (17.54%), astrocytomas (5.25%), ependymomas (1.75%), and embryonal tumors (1.75%). The remaining 10.53% corresponded to other types of tumors, including vestibular schwannomas and brain metastases.
Hematologic information
The blood groups were O 66.67%, A 29.82%, B 1.75%, and AB 1.75%. Rh-positivity was found in 96.49% of patients and negative in 3.51% of patients. O + blood type was 66.67%, followed by A + 30.91%, B + 1.82% and AB + 1.82%. Variations in static coagulation tests were found in 28.07% of the samples, mostly corresponding to the alteration of fibrinogen value. Patients with normal coagulation profile had a mean fibrinogen of 257.13 milligrams/deciliter, those with hypercoagulable profile had a mean of 288.72 milligrams/ deciliter and those with hypocoagulable profile had a mean of 284.33 milligrams/deciliter. The thromboelastogram used as a dynamic coagulation test was normal in 56.14% (32 subjects) and altered in 43.86% (25 subjects): 38.60% with a hypercoagulable profile (22 subjects) and 5.26% with a hypocoagulable profile (3 subjects). When thromboelastogram alterations were evaluated according to sex, an alteration was found in 29.17% of men (7/24), whereas 70.83% were normal (14/24). Regarding females, 54.55% had alterations in the thromboelastogram (18/33), and 45.45% did not (15/33). It was found that 29.17% of men and 54.55% of women had some type of alteration in the thromboelastogram.
| Table 1. Characterization of the study population | ||
|---|---|---|
| Variable | Measure of central tendency | Dispersion measures |
| Age | 48.63 years ¥ | 15.28 § |
| Sex | ||
| Masculine, n (%) | 24 (42.11) | – |
| Feminine, n (%) | 33 (57.89) | – |
| Weight | 68.66 kg ¥ | 10.36 § |
| Size | 166.08 cm ¥ | 8.89 § |
| Tumor type | ||
| Benign | 29 | 50.88 § |
| Malignant | 28 | 49.12 § |
| Histology | ||
| Astrocytoma n (%) | 3 (5.26) | – |
| Ependymoma n (%) | 1 (1.75) | – |
| Meningioma n (%) | 19 (33.33) | – |
| Embryonal tumors n (%) | 1 (1.75) | – |
| Glioblastoma n (%) | 10 (17.54) | – |
| Pituitary n (%) | 17 (29.82) | – |
| Other n (%) | 6 (10.53) | – |
| Midline deviation | ||
| < 5 mm, n (%) | 40 (29.82) | – |
| > 5 mm, n (%) | 17 (70.18) | – |
| BMI | 24.5 kg/m2^ | 22.8 – 26.3 ± |
¥: media; ^: median; §: Standard deviation (SD); ±: Interquartile range (IQR).
There was a higher prevalence of initial TEG alterations in females (54.55%) than in males (29.17%); however, this difference was not statistically significant (P = 0.057). Regarding the alteration of the static and dynamic coagulation tests, 8.77% of the patients (5/57) presented alterations in both tests, while 28.07% (16/57) had alterations only in the static tests. The data from the static and dynamic tests are presented in Table 2. Intraoperative variables: Median surgical bleeding was found to be 200 ml (RIQ 100-400 ml). However, the range of bleeding was quite wide, being found in the lowest percentile 30 mL and 99th percentile (2,500 mL); the latter during the resection of a vestibular schwannoma. Benign lesions showed a higher median bleeding volume than malignant lesions (455 mL vs. 293 mL); this difference was not statistically significant. There was a trend for higher bleeding in hypercoagulable patients than in hypocoagulable patients (440 mL vs. 333 mL) or those with a normal coagulation profile (336 mL), as illustrated in Table 3.
The requirement for transfusion of packed red blood cells in the sample was 10.52% (6 subjects). Only 1.75% (1 subject) required fresh frozen plasma and platelets. Data for each outcome are presented in Table 2. Postoperative outcomes: During the 30day follow-up of patients after brain tumor resection, 7.02% of the subjects required reintervention. The mean number of days of intensive care unit (ICU) stay was 3.26 and the mean number of days of hospital stay was 4.82 days. Patients with a normal coagulation profile had a mean ICU stay of 3.03 days, hypercoagulable patients had a higher mean of 3.72 days, and those with a hypocoagulable profile had the lowest mean number of days in the unit, 2.33 days. Therefore, no significant difference in the ICU length of stay was found. Only one patient (1.75%), a 45-year-old female with a pituitary adenoma, with no alteration in the coagulation profile, died during the follow-up period following the need for surgical reintervention. None of the patients presented with thrombotic complications during the follow-up. No patient presented with acute myocardial infarction or transfusion requirements during monitoring in the intensive care unit.
Bivariate analysis was performed, taking the alteration in the patients’ coagulation profile as an outcome.
| Table 2. Static and dynamic coagulation tests and intraoperative and postoperative outcomes in the population | ||
|---|---|---|
| Variable | Median | Standard deviation |
| Static tests | ||
| Hemoglobin | 14.08 | 1.82 |
| Platelets | 274.491.2 | 66001.66 |
| Fibrinogen | 270.75 | 104.79 |
| Partial thromboplastin time (PTT) | 27.66 | 3.23 |
| TTP control | 26.91 | 1.26 |
| Prothrombin time (PT) | 11.24 | 1.61 |
| PT Control | 11.00 | 0.51 |
| INR | 1.00 | 0.08 |
| Alteration of static tests | ||
| Yes n (%) | 16 (28.07) | – |
| No n (%) | 41 (71.93) | – |
| Initial TEG | ||
| R | 4.68 | 3.25 |
| K | 1.85 | 1.03 |
| Alpha angle | 62.53 | 12.39 |
| MA | 68.25 | 7.05 |
| G | 11.53 | 3.75 |
| LY30 | 0.24 | 1.21 |
| IC | 1.66 | 3.19 |
| Alteration TEG | ||
| Yes n (%) | 24 (43.86) | – |
| No n (%) | 32 (56.14) | – |
| Transfusion of hemocomponents | ||
| Red blood cells | 658 mL | 284.01 |
| Cryoprecipitates | – | – |
| Fresh frozen plasma | 600 mL | – |
| Platelets | 250 mL | – |
| Reintervention | ||
| Yes n (%) | 4 (7.02) | – |
| No n (%) | 53 (92.98) | – |
| Death within 30 days | ||
| Yes n (%) | 1 (1.75) | – |
| No n (%) | 56 (98.25) | – |
| UCI stay | 3.26 días | 2.09 |
| Hospital stay | 4.82 días | 3.07 |
| Thromboembolic complications | ||
| Yes n (%) | 0 (0) | – |
| No n (%) | 57 (100) | – |
| Surgical bleeding | 200 mL fl | 100 – 400 ± |
ÏÏ mediana; ±: Interquartile range (IQR).
Discussion
With the development of the present study, we obtained the first characterization of the type of brain neoplasm experienced by patients treated in a fourth-level Colombian hospital. Most of the subjects had benign tumors, with meningiomas being the most frequent lesions, followed by pituitary tumors and glioblastomas. In oncological patients, there is a hypercoagulable or prothrombotic state secondary to the capacity of tumor cells to activate the coagulation system, which leads to a significant percentage of mortality and morbidity (up to 50 %) in cancer patients[11],[12].
| Table 3. Characterization of the population according to coagulation profile | |||
|---|---|---|---|
| Variable | Hypocoagulable profile | Normal profile | Hypercoagulable profile |
| Age (years) | 63 | 44.5 | 48 |
| Sex (n) | |||
| Feminine | 2 | 15 | 16 |
| Masculine | 1 | 17 | 6 |
| UCI stay (days) ¥ | 2.33 | 3.03 | 3.72 |
| Hospital stay (days) ¥ | 3.66 | 4.56 | 5.36 |
| Initial TEG ¥ | |||
| R | 14.13 | 4.95 | 3.00 |
| K | 4.5 | 1.99 | 1.29 |
| Alpha angle | 39.16 | 62.16 | 66.25 |
| MA | 64.1 | 65.46 | 72.88 |
| G | 9.2 | 9.99 | 14.10 |
| LY30 | 0.0 | 0.09 | 0.5 |
| IC | -7.86 | 0.85 | 4.14 |
| Fibrinogen ¥ | 284.33 | 257.13 | 288.72 |
| Bleeding (mL) ^ | 333.33 | 336.25 | 439.54 |
| Histology | |||
| Astrocytoma | 0 | 2 | 1 |
| Ependymoma | 0 | 1 | 0 |
| Meningioma | 1 | 1 | 6 |
| Embryonal tumors | 0 | 0 | 1 |
| Glioblastoma | 1 | 7 | 2 |
| Pituitary | 1 | 8 | 8 |
| Other | 0 | 2 | 4 |
¥: media, ^: median.
When comparing the coagulation profiles of the subjects in our study with the data reported by Nates and collaborators[13], a greater alteration was found (56.14 % vs. 18%). Hypercoagulable subjects had a higher prevalence (36.60% vs. 9%); however, in the hypocoagulable group, the incidence was lower (5.26% vs. 9%). The proportion of alterations in the coagulation profile found in our study was higher than that reported by Nates et al., in 2007[13]. This indicates different demographics with discordant patterns of perioperative coagulation disorders between the two populations.
The main malignant tumor characterized in our study was glioblastoma multiforme, which chronically activates the coagulation system, producing intratumoral thrombosis by the emission of procoagulant agents, and is also associated with spontaneous or iatrogenic bleeding[4]; therefore, malignant brain tumors are expected to bleed to a greater extent. In particular, the results of this study do not support this hypothesis because subjects with benign tumors bled more than those with malignant tumors.
Traditionally, perioperative patients are evaluated using static coagulation tests that monitor both the extrinsic and intrinsic pathways of the coagulation cascade, fibrinogen activity, and platelet count. However, TEG, which is part of the dynamic viscoelastic coagulation test and measures the coagulation process in real time by clot development, allowing early detection of hemostatic disorders[14], is not frequently used in the neurosurgical patient setting.
According to the results obtained, there was no concordance between the static tests and the initial thromboelastogram; thus, the performance of the hematological screening could be optimized with viscoelastic tests to the extent that this type of resource is available, since it detected 25 alterations in 57 subjects, while the static tests detected only 16 of these 25 altered subjects in the thromboelastogram.
Patients with a hypocoagulable-type coagulation profile were expected to have lower fibrinogen levels than patients who were hypercoagulable because this protein is associated with blood clot formation[15]; however, patients with alterations in the initial hypocoagulable-type coagulation profile were found to have higher fibrinogen levels than patients who were hypercoagulable or had a normal coagulation profile.
Patients with a hypocoagulable-type coagulation profile are expected to have higher intraoperative bleeding than those with a hypercoagulable profile and are not on disseminated intravascular coagulation. However, it is relevant to mention that in our study, subjects with hypercoagulable coagulation profiles bled to a greater extent than those with hypocoagulable profiles. In relation to ICU stay during the immediate postoperative period, our results agree with those reported by the Colombian Association of Critical Medicine and Intensive Care in 2023 (3.26 VS 3 vs. 4 days)[16].
One of the main strengths of the present study is that, to date, this is the first report of coagulation alterations in patients undergoing surgical resection of brain tumors with histopathological characterization of intracranial neoplasms associated with their main outcomes in the first postoperative month. Additionally, patients undergoing brain tumor resection were managed according to standardized management. One of the limitations is the sample size, since the number of patients included in the present cohort, in contrast to the specificity of the population and the low prevalence of the outcomes, could mean that, in many cases, the absence of statistical significance may be due to a lack of power.
From the present study, it can be concluded that although TEG as a dynamic coagulation test has been shown to be an effective method for assessing coagulation status in multiple types of patients, including neurological patients, no relationship was observed between coagulation patterns obtained from the thromboelastogram and perioperative outcomes, such as hospital stay, intensive care unit stay, and mortality. In addition, TEG was more efficient in identifying altered states of the coagulation profile compared to static coagulation testing, confirming that, in centers where viscoelastic testing is available, patients would benefit more from this type of dynamic testing preoperatively than from routine static coagulation testing, and we found that the proportion of women undergoing surgical resection of brain tumors with coagulation abnormalities measured by TEG was higher than that of men. Although this was not a statistically significant difference, it may generate associated clinical hypotheses.
Patients undergoing resection of benign brain tumors whose coagulation profile classifies them as hypercoagulable present with greater intraoperative bleeding, which is why additional measures should be taken in this type of patient, such as having a reserve of hemocomponents or blood-saving strategies.
Referencias
1. NIH National Cancer Institute. Importance of cancer research and its effect on public health [Internet]. [cited 2022 Sep 20]. Available from: https://www.cancer.gov/espanol/investigacion/areas/salud-publica
2. Ministerio de Salud y Protección Social. Salud Pública, Prevención de enfermedades no transmisibles, Cáncer [Internet]. [cited 2022 Sep 20]. Available from: https://www.minsalud.gov.co/salud/pub- lica/PENT/Paginas/Prevenciondel-cancer.aspx
3. Ferlay J, Ervik M, Lam F, Laversanne M, Colombet M, Mery L, et al. Global Cancer Observatory: Cancer Today. Lyon, France: Inter- national Agency for Research on Cancer; 2022.[Internet][ [cited 2024 Nov 7]], Available from https://gco.iarc.who.int/today
4. Magnus N, D’Asti E, Garnier D, Meehan B, Rak J. Brain neoplasms and coagulation. Semin Thromb Hemost. 2013 Nov;39(8):881-95. https://doi.org/10.1055/s-0033-1357483 PMID:24108471
5. Mathews M, Newman R, Chappell ET. Management of coagulopathy in the setting of acute neurosurgical disease and injury. Neurocrit Care. 2006;5(2):141-52. https://doi.org/10.1385/NCC:5:2:141 PMID:17099261
6. Shi S, Cheng J, Zhao Y, Chen W. Incidence, and preoperative and intraoperative prognostic factors of deep venous thrombosis in patients with glioma following craniotomy. Clin Neurol Neu- rosurg. 2021 Nov;210:106998. https://doi.org/10.1016/j.clineuro.2021.106998 PMID:34739883
7. Mandoj C, Tomao L, Conti L. Coagulation in Brain Tumors: Biological Basis and Clinical Implications. Volume 10. Frontiers in Neurology. Frontiers Media S.A.; 2019.
8. Heesen M, Kemkes-Matthes B, Deinsberger W, Boldt J, Matthes KJ. Coagulation alterations in patients undergoing elective craniotomy. Surgical Neurology [Internet]. 1997 Jan;47(1):35–8. Available from: http://dx.doi.org/10.1016/s0090-3019(96)00388-6
9. Ganter MT, Hofer CK. Coagulation monitoring: current techniques and clinical use of viscoelastic point-of-care coagulation devices. Anesth Analg. 2008 May;106(5):1366-75. https://doi.org/10.1213/ane.0b013e318168b367 PMID:18420846
10. Kvint S, Schuster J, Kumar MA. Neurosurgical applications of viscoelastic hemostatic assays. Neurosurg Focus. 2017 Nov;43(5):E9. https://doi.org/10.3171/2017.8.FOCUS17447 PMID:29088950
11. Caine GJ, Stonelake PS, Lip GY, Kehoe ST. The hypercoagulable state of malignancy: pathogenesis and current debate. Neoplasia. 2002;4(6):465-73. https://doi.org/10.1038/sj.neo.7900263 PMID:12407439
12. Sosa-Quintero LS, Carrasco-Martínez IL, Mariscal-Ramírez I, García-Luna EE, Nava-Zavala AH, Rubio-Jurado B. The protrombotic state in patients with cancer. Volume 20. Gaceta Mexicana de Oncologia. Publicaciones Permanyer; 2021. pp. 26-34.
13. Nates JL, Aravindan N, Hirsch-Ginsberg C, Sizer KC, Kee S, Nguyen AT, et al. Critically ill cancer patients are not consistently hypercoagulable after craniotomy. Neurocrit Care. 2007;7(3):211-6. https://doi.org/10.1007/s12028-007-0064-2 PMID:17968522
14. Ko RH, Ji L, Young G. A novel approach for detecting hypercoagulability utilizing thromboelastography. Thromb Res. 2013 Apr;131(4):352-6. https://doi.org/10.1016/j.throm- res.2013.01.031 PMID:23419411
15. Nielsen VG, Lemole GM Jr, Matika RW, Weinand ME, Hussaini S, Baaj AA, et al. Brain tumors enhance plasmatic coagulation: the role of hemeoxygenase-1. Anesth Analg. 2014 May;118(5):919- 24. https://doi.org/10.1213/ANE.0000000000000048 PMID:24413553
16. Naranjo Ramírez AF. Medrano Areiza Á de J, Arango Sánchez B, Arango Martínez JC, Naranjo Atehortúa LF. Postoperative care of brain tumor surgery in critical care unit. Vol. 24, Acta Colombiana de Cuidado Intensivo. Asociacion Colombiana de Medicina Critica y Cuidado lntensivo; 2024. p. 140-51.

 ORCID
ORCID