Sameh M. Hakim1, Hanaa A. Elgendy1, Doaa M. Kamal El-Din2, Mohamed M. Rashed2, Ghada R. Elgiushy3, Mohamed O. Elgouhary2
Recibido:
Aceptado:
©2026 El(los) Autor(es) – Esta publicación es Órgano oficial de la Sociedad de Anestesiología de Chile
Revista Chilena de Anestesia Vol. 55 Núm. 2 |https://doi.org/10.25237/revchilanestv55n2-08
PDF
Estimación de la longitud para colocar la punta del catéter venoso central en el punto medio de la vena cava superior
Abstract
Purpose: This study aimed to derive a prediction rule for estimating the required length to place the tip of a right internal jugular vein (IJV) catheter at the mid-point of the superior vena cava using surface landmarks. Methods: The distance from the apex of Sedillot’s triangle to the ipsilateral sternoclavicular joint and from the latter to the lower border of the right second sternocostal junction was summed and regressed on the actual length required to place the catheter tip at 30 mm cephalad to the atrio-caval junction as determined with transesophageal echocardiography (TEE) in 70 adults undergoing right IJV catheterization. The accuracy of the proposed rule was evaluated by calculation of the standard error of the estimate (SEest) and was compared with Peres’ formula and the C-length methods. Results: The proposed rule had a SEest of 8.4 mm compared with 14.0 mm and 30.7 mm for the C-length and Peres’ formula, respectively. The standardized mean difference (95% confidence interval) between the catheter length predicted with the surface landmarks rule and that estimated with TEE was 0.00 (-0.24, 0.24), P > .999. The corresponding difference from the TEE estimations was -0.36 (-0.61, -0.12), P = .003 and 2.42 (2.02, 2.82), P < .0001, for the C-length and Peres’ formula, respectively. K-fold cross-validation of the proposed rule showed a mean cross-validation score of 0.135 mm. Conclusion: The proposed surface landmarks rule has a high accuracy that is comparable to TEE and performs better than Peres’ formula or the C-length method. External validation of the rule is required.
Resumen
Purpose: This study aimed to derive a prediction rule for estimating the required length to place the tip of a right internal jugular vein (IJV) catheter at the mid-point of the superior vena cava using surface landmarks. Methods: The distance from the apex of Sedillot’s triangle to the ipsilateral sternoclavicular joint and from the latter to the lower border of the right second sternocostal junction was summed and regressed on the actual length required to place the catheter tip at 30 mm cephalad to the atrio-caval junction as determined with transesophageal echocardiography (TEE) in 70 adults undergoing right IJV catheterization. The accuracy of the proposed rule was evaluated by calculation of the standard error of the estimate (SEest) and was compared with Peres’ formula and the C-length methods. Results: The proposed rule had a SEest of 8.4 mm compared with 14.0 mm and 30.7 mm for the C-length and Peres’ formula, respectively. The standardized mean difference (95% confidence interval) between the catheter length predicted with the surface landmarks rule and that estimated with TEE was 0.00 (-0.24, 0.24), P > .999. The corresponding difference from the TEE estimations was -0.36 (-0.61, -0.12), P = .003 and 2.42 (2.02, 2.82), P < .0001, for the C-length and Peres’ formula, respectively. K-fold cross-validation of the proposed rule showed a mean cross-validation score of 0.135 mm. Conclusion: The proposed surface landmarks rule has a high accuracy that is comparable to TEE and performs better than Peres’ formula or the C-length method. External validation of the rule is required.
Introduction
The internal jugular vein (IJV) is the preferred route for central venous cannulation when it is anticipated that a central venous catheter (CVC) would be in use for a limited period, as is the case in perioperative insertions, given its reputably lower risk for CVC-related mechanical complications[1]. The right IJV is normally the preferred choice for cannulation owing to its straight course to the superior vena cava (SVC), which helps place the catheter within the IJV or the SVC in a parallel orientation to the wall of the great veins, thus reducing the risk of perforation and cardiac tamponade[2].
Central venous cannulation with the advancement of the catheter to an empirical length ends in approximately one-quarter of the catheters placed in the right atrium (RA)[3]. In this regard, placement of a CVC tip in the RA or the lower SVC carries a potential risk of cardiac tamponade[4], which, though a rare event, is associated with a dreadfully high mortality rate in the order of 65%[5]. Notably, the Food and Drug Administration recommended that, except for pulmonary artery catheters, no CVC be introduced into the cardiac cavity[6].
A cadaveric study showed that the pericardial reflection extended for three centimeters on the medial side of the SVC and that the carina was situated below the pericardial reflection in all the cadavers they studied. The investigators concluded that a CVC tip had to be placed above the carina to negate the risk of cardiac tamponade in case erosion of the venous wall occurred[7]. However, data from in vivo studies have questioned the validity of this recommendation. For instance, a radiological study showed that the upper limit of the pericardial reflection was approximately at the mid-point of the SVC and was consistently seen in the same computed tomography slices as the takeoff of the right mainstem bronchus. The carina, on the other hand, was situated caudad to the pericardial reflection by a mean of 0.7 cm and caudad to the mid-point of the SVC by a mean of 1.3 cm. Therefore, the authors suggested using the right main bronchus takeoff as a more reliable landmark to minimize the risk of tamponade[8].
In the present trial, we attempted to derive a prediction rule to estimate the required length for placement of the CVC tip at the mid-point of the SVC from simple anatomical landmarks that are consistent with the known surface projections of the great veins. The accuracy of this rule was examined using TEE and was intended for adult patients undergoing cannulation of the right IJV utilizing a middle approach. As a secondary aim, we examined the performance of this suggested rule as contrasted with two other popular methods for estimation of the CVC length, the Peres formula[3] and the C-length method[9].
Patients and Methods
This single-center, prospective, clinical study was conducted during the period from September 2019 to December 2020. The study protocol was approved by the Institutional Review Board and was registered at the ClinicalTrials.gov Protocol Registry. Written informed consent was obtained from all participants, and the trial was monitored by the local institutional Data and Safety Monitoring Board.
Patient selection
Patients > 18 years of age of either sex scheduled for elective cardiac surgery in whom TEE was indicated or recommended were eligible. Exclusion criteria included previous cardiothoracic or neck surgery, previous radiotherapy on the neck or chest, intrathoracic or neck mass lesions, deformity of the chest wall or cervicothoracic spine, or congenital anomalies of the heart or great vessels. All patients were planned for central venous cannulation utilizing a middle approach to the right IJV.
Interventions
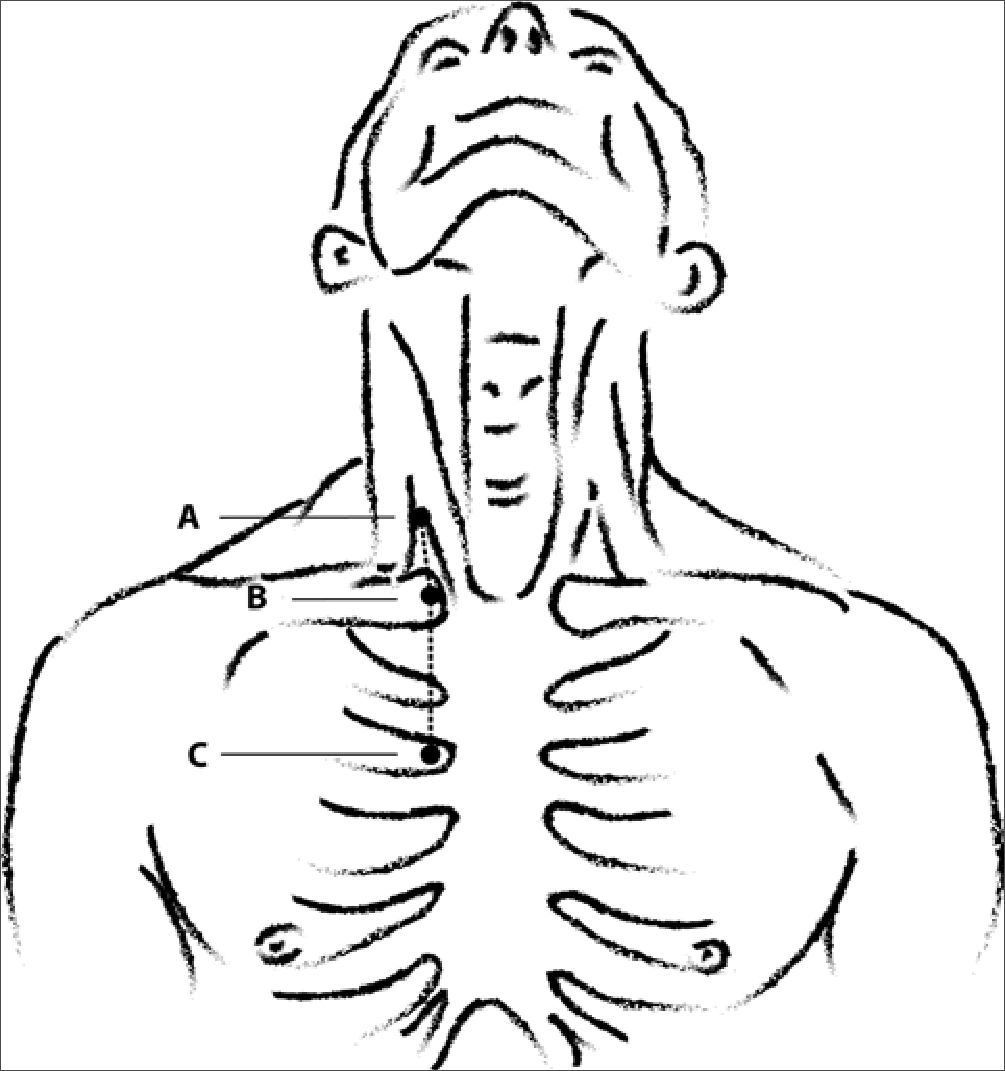
Before induction of anesthesia, three sets of measurements were obtained on each patient: 1) Surface distance: This measure was obtained by placing three points on the skin using a non-erasable marker, point A at the apex of the triangle between the two heads of the sternomastoid (Sedillot’s triangle), point B at the ipsilateral sternoclavicular joint, and point C at the lower border of the right second sternocostal junction. The distance between point A and point B and between point B and point C was measured in millimeters and summed (Figure 1)[10]; 2) C-length: Using the Picture Archiving and Communications System (PACS) Synapse® 6, software version 4.4.3 (FUJIFILM Medical Systems, Lexington, MA, USA), the distance from the tip of the transverse process of the first thoracic vertebra (T1) to the tracheal bifurcation (carina of the lung) was measured in millimeters[9]; 3) Peres’ formula: According to Peres’ formula, the patient’s height in centimeters was divided by ten to determine the CVC length expected to have the CVC tip placed in the SVC. The expected CVC length was then converted to millimeters[3]. Preoperative measurements were taken by one of two cardiac anesthesiologists not involved in IJV cannulation or the conduct of TEE examination.
Anesthesia was induced and maintained according to the standard institutional protocols. A TEE probe (GE Vivid 6®, GE Healthcare®, Wauwatosa, WI, USA) was introduced, and the multiplane angle was adjusted between 80° and 110° until a clear bicaval view was obtained to visualize the caudad part of the SVC at its entry into the RA. The TEE probe was then manipulated to display the SVC as far as possible from its junction with the RA. TEE examinations were conducted by one of two cardiac anesthesiologists certified in perioperative TEE.
Figure 1. Hand-drawn art line illustrating the proposed surface landmarks method. Point A: Apex of Sedillot’s triangle (triangular space between the sternal and clavicular heads of the sternomastoid muscle); Point B: Sternoclavicular joint; Point C: Lower border of second sternocostal junction. The distance between point A and point B and between point B and point C is measured in millimeters and summed. Adapted by the authors from Cunningham DJ[10]. The original illustration is in the public domain.
Unless contraindicated, the patients were then placed in a 15to 20-degree head-down (Trendelenburg) position with a slight leftward rotation of the head, and the skin was sterilized with povidone-iodine and draped. The right IJV was cannulated under ultrasound guidance using the Seldinger technique and utilizing a middle approach to the vein. Once the vein was cannulated, the syringe was disconnected, and a J-wire was advanced through the trocar until the J-end of the guide wire was visualized at the SVC-RA junction via the TEE. The trocar was withdrawn from the skin, and the dilator was passed over the guide wire to dilate the passage of the CVC through the skin and subcutaneous tissue. The CVC was then threaded over the guide wire until the tip was seen at the atrio-caval junction. The guide wire was removed, and the CVC was withdrawn for 30 mm as measured with a sterile flexible ruler. The length of the exteriorized portion of the CVC was determined with the flexible ruler, and the measured length was subtracted from the total length of the catheter (20 cm) to calculate the length of the inserted portion of the CVC with the tip placed at the mid-SVC. Cannulations of the IJV and adjustment of the CVC tip at the desired depth were carried out by the same anesthesiologist.
After surgery, all patients were transferred to the cardiac surgical intensive care unit (ICU), where a portable anteroposterior (AP) chest radiograph was obtained. To ensure consistency in assessments, all images were acquired using a standardized protocol. Patients were propped up in a 60-degree head-up (semi-Fowler’s) position with the head and neck centralized in the neutral position, in alignment with the trunk and limbs. If not able to maintain the desired position, an assistant helped stabilize the patients during the acquisition of the image. Images were acquired at end-expiration with attention to radiographic quality, including exposure, rotation, and adequacy of lung inflation. Radiographs were reviewed at full resolution on the PACS, and brightness/contrast adjustments were permitted to enhance visualization of anatomical structures and catheter course. The projection was deemed optimal if the carina and main bronchi were readily visible with the trachea in the midline and the catheter tip identified. Any films deemed suboptimal in quality were repeated. The level of the right main bronchus takeoff was taken as the anatomical reference for CVC tip depth, and the distance of the CVC tip from that line was measured in millimeters. Accordingly, distances cephalad to the reference line were identified arithmetically with a minus (-) sign and distances caudad to it with a plus (+) sign, to distinguish mathematically whether the CVC tip was placed short of or beyond the reference line, respectively. The proportion of patients associated with each of the three rules in whom the CVC tip would be virtually placed at or cephalad to the reference line was determined. For this purpose, we added the actual distance between the CVC tip and the reference line to the estimated CVC length as determined with the predictive rule, then we subtracted the inserted length of the CVC as guided with TEE from that value. All chest radiographs were rated by a single anesthesiologist with over 15 years of experience in radiological assessment of CVC imaging. The rater was not involved in the preoperative or intraoperative care of the patients and was blinded to other clinical details.
Outcome measures
The primary outcome measure was the accuracy of the three prediction rules (i.e., the surface landmarks-based rule, C-length, and Peres’ formula) for estimation of the CVC length required to place the CVC tip in the mid-SVC using TEE as the gold standard. The secondary outcome measures were: 1) the degree of agreement between each of the three non-invasive rules and TEE, and 2) the proportion of patients associated with each of the three rules in whom the CVC tip would be placed at or cephalad to the right main bronchus takeoff line on postoperative chest radiographs.
Statistical methods
Sample size calculation
The required sample size was calculated using the Power Analysis and Sample Size Software© (PASS©) v. 11.0.8 (NCSS, LLC, Kaysville, Utah, USA, ncss.com/software/pass). We planned to regress the actual length required to place the CVC tip in the mid-SVC, as determined with TEE guidance (outcome variable), on the expected length as estimated non-invasively from the surface landmarks (explanatory variable). Since we had no prior information regarding the estimates of such an equation, we identified a minimum coefficient of determination (R2) of .25 as a clinically important goodness-of-fit measure for the proposed prediction rule. This was based on the suggestion that an R2 of >.15 or more would indicate a meaningfully good fit for clinical models [11]. So, we calculated that a sample size of 55 patients would achieve a power of 99% to detect a non-zero R2 attributed to one explanatory variable (surface distance) using a two-sided t-test. The actual value of R2 was assumed to equal .25, and the significance of the test was targeted at a conventional value of P < .05. Expecting a drop-out rate of no more than 20%, we recruited 70 patients to provide for sample attrition.
Statistical methods
Data were analyzed using the MedCalc® Statistical Software version 22.009 (MedCalc® Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2023). Continuous numerical variables were presented as mean and standard deviation (SD), and categorical variables as number and percentage.
To derive a prediction rule based on the proposed landmarks method, we used a simple linear regression model wherein we regressed the actual length needed to place the CVC tip at the mid-SVC, as guided with TEE (dependent variable), on the expected length as estimated with the surface landmarks method (independent variable). The accuracy of the prediction rule was assessed by calculation of the standard error of the estimate (SEest), which is the SD of the residuals. For the C-length method and Peres’ formula, we determined the SEest by calculating the difference between the expected CVC length and the actual length for each patient. Then, we squared the differences, summed them, and calculated the mean squared difference. The SEest was taken as the square root of the mean squared difference (SEest = Vmeannsquared difference) K-fold cross-validation of the proposed rule was done utilizing the Pandas [12] and Scikit-learn[13] libraries in Python version 3.12.0 (October 2023)[14].
The inter-method agreement was examined using Bland-Altman plots[15],[16].
Related numerical data were compared using repeated measures analysis of variance with application of the Holm-Bonferroni correction for post-hoc pairwise comparisons. The Cochran Q test was used to compare related binary data, and post-hoc comparisons were done using the McNemar test with application of the Holm-Bonferroni correction. The Sheskin method was used to calculate the minimum required difference for related binary data. Differences are reported as both standardized (Cohen’s dz) and raw unstandardized (raw) differences with a 95% confidence interval (95% CI). The Wilson method was used to calculate the 95% CI for binary data, as it is more robust for near-extreme percentages. For continuous data, the standard t-distribution method was used. All reported P values are two-sided. P values < .05 are considered statistically significant.
Results
The present study included 70 patients, 35 (50%) males and 35 (50%) females, with a mean ± SD age of 49 ± 14 years (range, 18 to 85 years). The characteristics of the study population are summarized in Table 1. The details of regressing the TEE-verified length needed to place the CVC tip at the mid-SVC on the measured surface distance, including the goodness-offit tests, are shown in Supplementary Digital Material 1. Supplementary Digital Materials 2-4 show the scatter plots for testing the assumptions of linear regression. The derived regression equation was as follows:
Expected CVC length in mm = 64.919+(0.516* Measured surface distance in mm)
Primary outcome
The proposed surface landmarks rule had a SEest of 8.4 mm compared with 14.0 mm and 30.7 mm for the C-length and Peres’ formula, respectively. The mean (95% CI) of the difference between the CVC length predicted with the surface landmarks rule and that estimated with TEE was 0.0 (-2.0, 2.0) mm (standardized mean difference [95% CI] = 0.00 [-0.24, 0.24], Holm-Bonferroni-corrected P value > .999). The corresponding difference (95% CI) from the TEE estimations was -4.8 (-8.0, -1.7) mm for the C-length (standardized mean difference [95% CI] = -0.36 [-0.61, -0.12], Holm-Bonferronicorrected P value = .003), and was 28.5 (25.7, 31.2) mm for Peres’ formula (standardized mean difference [95% CI] = 2.42 [2.02, 2.82], Holm-Bonferroni-corrected P value < .0001). We did k-fold cross-validation of the proposed rule using five equal splits of the original data, each including 14 observations. The cross-validation scores (root mean square errors) were -0.144, -0.218, 0.136, 0.443, and 0.460 mm, with a mean cross-validation score of 0.135 mm.
| Table 1. Characteristics of the study population | |
|---|---|
| Variable | Metric |
| Age (years) | 49 ± 14 (18, 85) |
| Male sex | 35 (50.0%) |
| Weight (kg) | 78 ± 15 (47, 122) |
| Height (cm) | 164 ± 8 (140, 180) |
| BMI (kg/m2) | 29.1 ± 5.2 (17.9, 46.7) |
| Surgery | |
| Aortic valve replacement | 8 (11.4%) |
| Bentall procedure | 1 (1.4%) |
| CABG | 26 (37.1%) |
| CABG plus aortic valve replacement | 2 (2.9%) |
| CABG plus mitral valve replacement | 3 (4.3%) |
| Mitral valve replacement | 19 (27.1%) |
| Mitral valve replacement plus tricuspid valve replacement | 2 (2.9%) |
| Pulmonary valve replacement plus perivalvular abscess drainage | 1 (1.4%) |
| Septal myomectomy | 2 (2.9%) |
| Septal myomectomy plus aortoplasty | 1 (1.4%) |
| Tricuspid valve replacement | 4 (5.7%) |
| VSD closure | 1 (1.4%) |
| Surface distance (mm) | 136 ± 13 (109, 169) |
| C-length (mm) | 131 ± 15 (95, 174) |
| Peres’ formula (mm) | 164 ± 8 (140, 180) |
| TEE-verified length needed to place CVC tip at mid-SVC (mm) | 135 ± 11 (110, 162) |
Data are presented as mean ± SD (minimum, maximum) or count (percentage). BMI = body mass index; CABG = coronary artery bypass grafting; CVC = central venous catheter; SVC = superior vena cava; TEE = transesophageal echocardiography; VSD = ventricular septal defect.
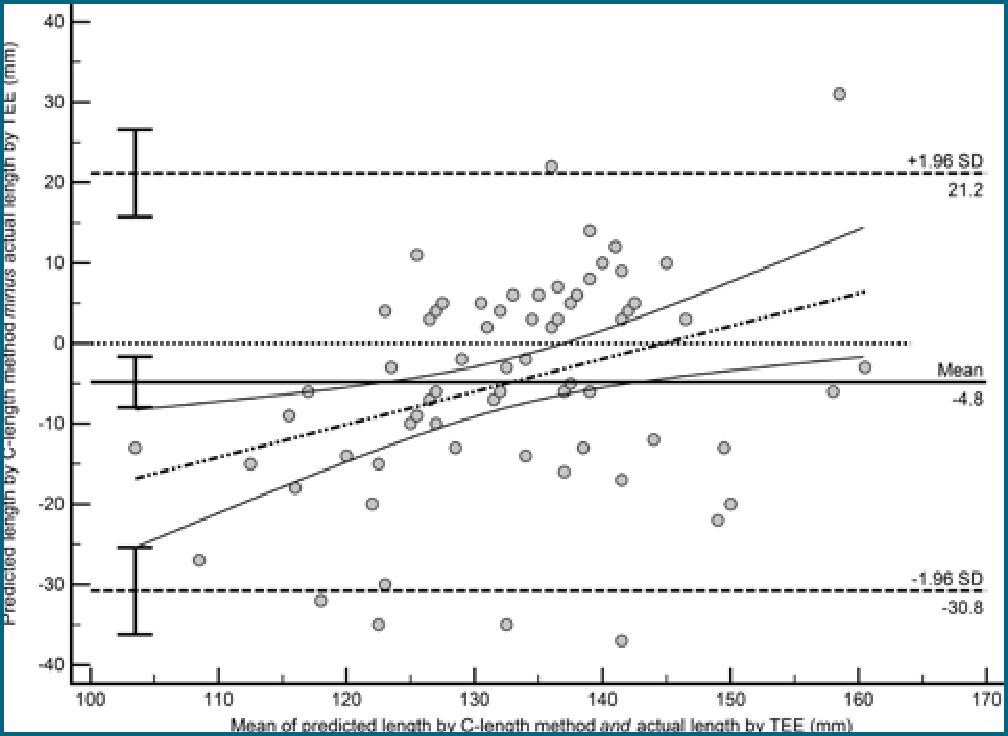
Figure 3. Bland-Altman plot showing the agreement between the C-length and transesophageal echocardiography. Lower limit of agreement (95% CI) = -30.8 (-36.2, -25.4) mm. Upper limit of agreement (95% CI) = 21.2 (15.7, 26.6) mm. Bias (95% CI) = -4.8 (-8.0, -1.7) mm.
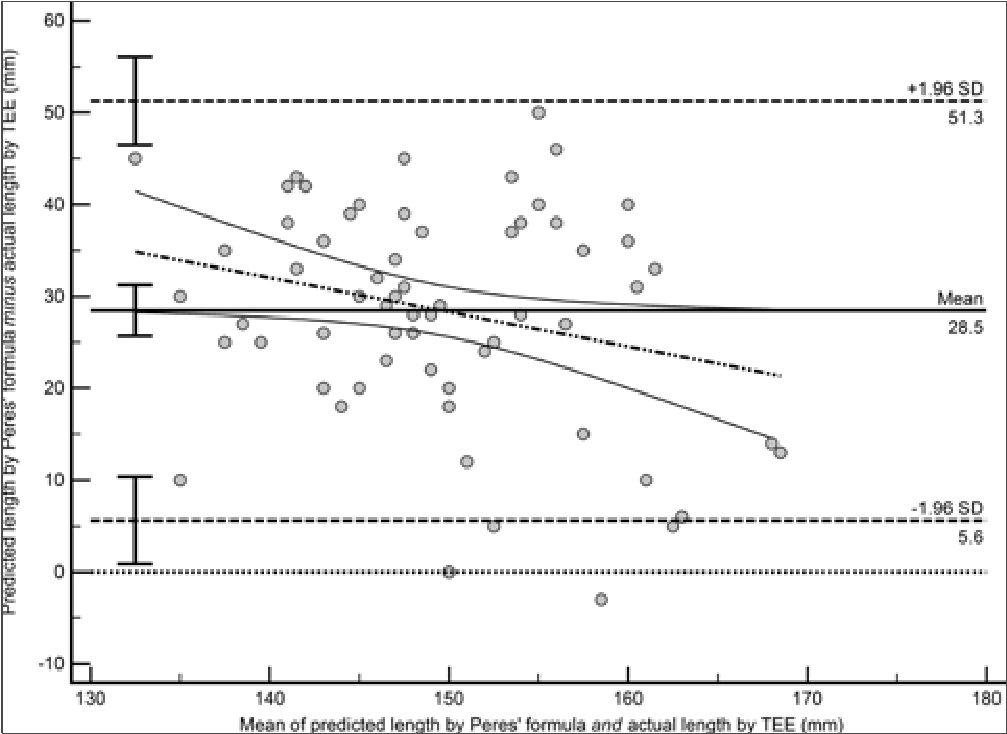
Figure 4. Bland-Altman plot showing the agreement between Peres’ formula and transesophageal echocardiography. Lower limit of agreement (95% CI) = 5.6 (0.9, 10.4) mm. Upper limit of agreement (95% CI) = 51.3 (46.5, 56.1) mm. Bias (95% CI) = 28.5 (25.7, 31.2) mm.
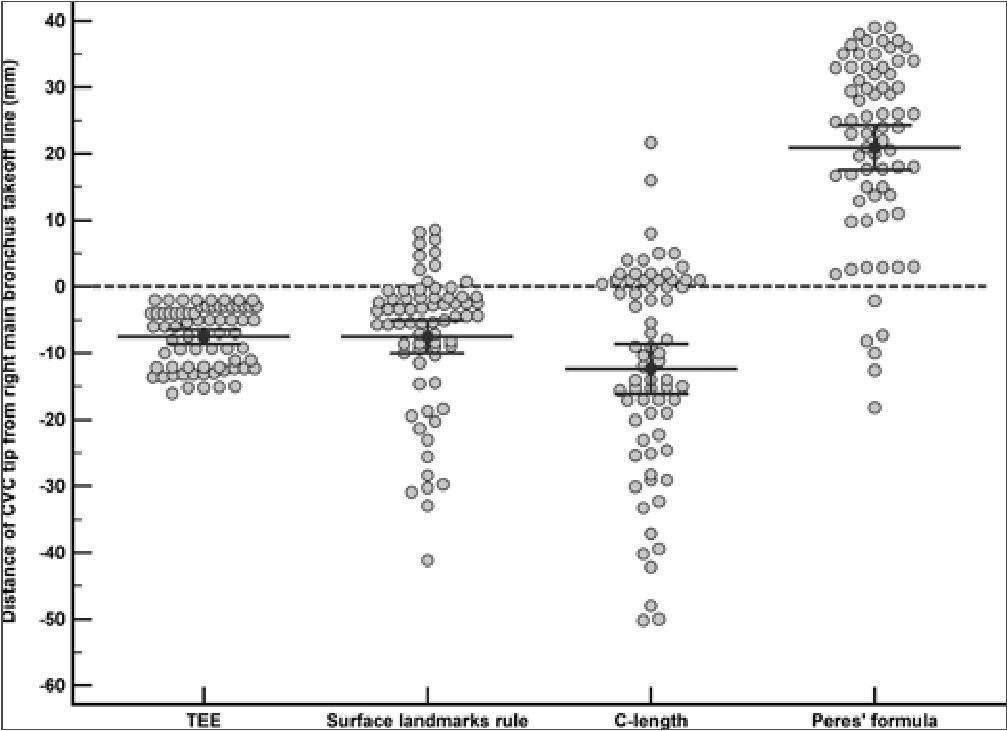
Figure 5. The distance between the CVC tip and the right main bronchus takeoff line as measured on postoperative chest radiographs for each of the four catheterization methods. Markers represent individual observations. Error bars represent the 95% confidence interval. The central line within the error bars represents the mean. The difference between the proposed surface landmarks rule and TEE was not statistically significant (Cohen’s dz [95% CI] = 0.00 [-0.24, 0.24], P value > .999). However, the C-length was associated with a shallower CVC tip location compared with TEE (Cohen’s dz = -0.43 [-0.73, -0.13], P value = .003) and Peres’ formula with a deeper CVC tip location compared with TEE (Cohen’s dz [95% CI] = 2.35 [1.96, 2.74], P value < .0001).
Discussion
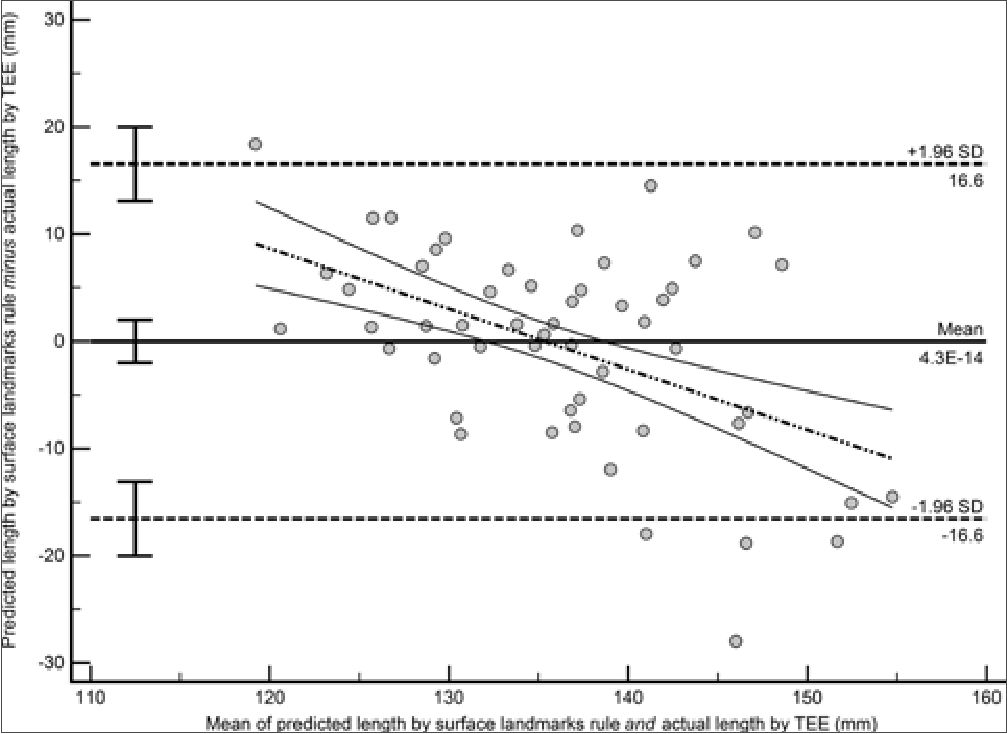
Figure 2. Bland-Altman plot showing the agreement between the proposed surface landmarks rule and transesophageal echocardiography. Lower limit of agreement (95% CI) = -16.6 (-20.0, -13.1) mm. Upper limit of agreement (95% CI) = 16.6 (13.1, 20.0) mm. Bias (95% CI) = 0.0 (-2.0, 2.0) mm.
Secondary outcomes
The limits of agreement between the proposed surface landmarks rule and TEE were from a lower limit of -16.6 (95% CI = -20.0 to -13.1) mm to an upper limit of 16.6 (95% CI = 13.1 to 20.0) mm, with a bias (mean difference) of 0.0 (95% CI = -2.0 to 2.0) mm (Figure 2), which was more acceptable clinically compared with the C-length (lower limit of agreement [95% CI] = -30.8 [-36.2, -25.4] mm, upper limit of agreement [95% CI] = 21.2 [15.7, 26.6] mm, bias [95% CI] = -4.8 [-8.0, -1.7] mm) (Figure 3) or Peres’ formula (lower limit of agreement [95% CI] = 5.6 [0.9, 10.4] mm, upper limit of agreement [95% CI] = 51.3 [46.5, 56.1] mm, bias [95% CI] = 28.5 [25.7, 31.2] mm) (Figure 4).
On postoperative chest radiographs, the mean (95% CI) of the actual distance between the CVC tip and the right main bronchus takeoff line was -7.5 (-8.6, -6.6) mm. The mean (95% CI) of the virtual distance between the hypothetical location of the CVC tip, as expected using the proposed surface landmarks rule, and the right main bronchus takeoff line was -7.5 (-10.0, -5.0) mm. The mean (95% CI) of the corresponding distances for the C-length and Peres’ formula were -12.3 (-16.1, -8.6) mm and 20.9 (17.6, 24.3) mm, respectively. The difference between the proposed surface landmarks rule and TEE was not statistically significant (mean difference [95% CI] = 0.0 [-2.0, 2.0] mm, Cohen’s dz [95% CI] = 0.00 [-0.24, 0.24], P value > .999). However, the C-length was associated with a shallower CVC tip location compared with TEE (mean difference [95% CI] = -4.8 [-8.0, -1.7] mm, Cohen’s dz = -0.43 [-0.73, -0.13], P value = .003). In contrast, Peres’ formula was associated with a deeper CVC tip location compared with TEE (mean difference [95% CI] = 28.5 [21.7, 27.2] mm, Cohen’s dz [95% CI] = 2.35 [1.96, 2.74], P value < .0001) (Table 2 and Figure 5).
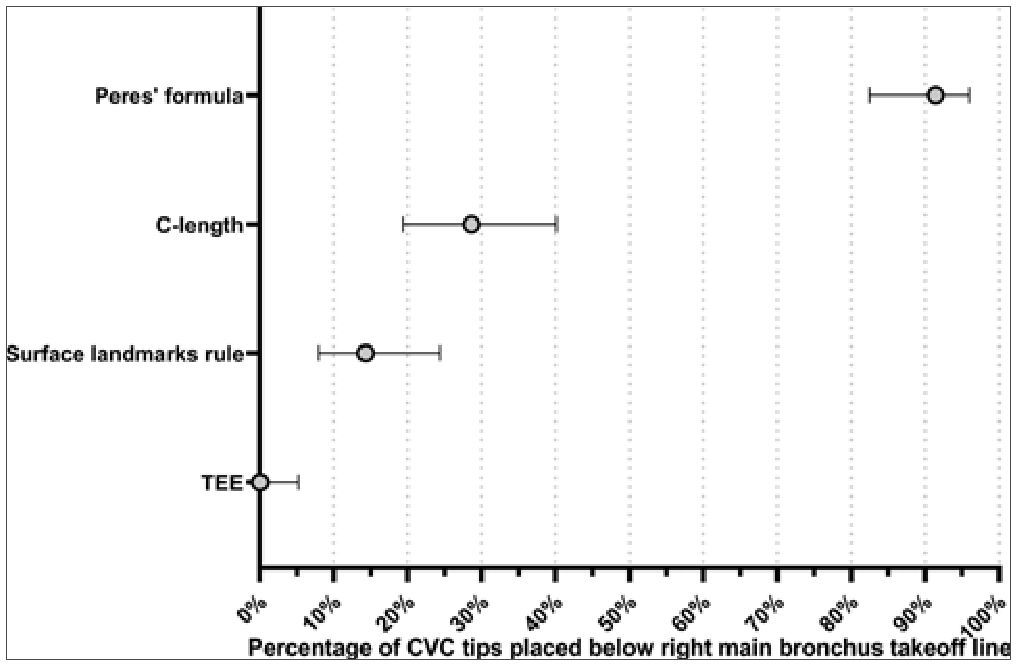
None of the patients (0.0%, 95% CI: 0.0% to 5.2%) had the CVC tip located caudad to the right main bronchus takeoff line on postoperative chest X-ray.The virtual location of the CVC tip, as estimated with the surface landmarks rule, C-length, or Peres’ formula, was caudad to the right main bronchus takeoff line in 10 (14.3%, 95% CI: 7.9% to 24.3%) patients, 20 (28.6%, 95% CI: 19.4% to 40.1%) patients, or 64 (91.4%, 95% CI: 82.5% to 96.0%) patients, respectively (Cochran Q X2[3] = 127.930, P value < .0001). The difference between the C-length and TEE (unstandardized difference [95% CI] = 28.6% [17.8%, 39.3%], Cohen’s dz [95% CI] = 0.63 [0.39, 0.87], P value < .0001), and between Peres’ formula and TEE (unstandardized difference [95% CI] = 91.4% [84.8%, 98.1%], Cohen’s dz [95% CI] = 3.24 [3.00, 3.24], P value < .0001), was statistically significant (Sheskin’s minimum required difference = 23.8%). In contrast, the difference between the proposed surface landmarks rule and TEE did not exceed Sheskin’s minimum required difference (unstandardized difference [95% CI] = 14.3% [6.1%, 22.5%], Cohen’s dz [95% CI] = 0.22 [0.09, 0.35], P value = .004, Sheskin’s minimum required difference = 23.8%) (Table 2 and Figure 6).
The present study showed that the proposed surface landmarks rule had high accuracy for the estimation of the length required to have the CVC tip placed at the mid-SVC for catheters inserted via the right IJV. The suggested rule could estimate the required length with an error of less than one centimeter. Compared with the C-length and Peres’ formula, the proposed rule showed better agreement with the TEE measurements and was associated with the least chance for placement of the CVC tip below the pericardial reflection.
Advancing CVCs to an empirical length is associated with an alarmingly high incidence of intra-atrial placement[3] and predisposes patients to cardiac tamponade that carries a dismal outcome, with about 80% of victims succumbing and 12% surviving in a vegetative state, according to one series[17].
On the other side, there is evidence that the farther the catheter tip is placed from the atrio-caval junction, the higher the risk of central venous thrombosis is[18]. An international consortium of experts made a Grade 1A recommendation (i.e., a strong recommendation based on a high level of evidence) that the tip of the CVC should be located at the confluence of the SVC with the RA to reduce the risk of catheter-related thrombosis[19]. However, despite the claimed benefit of that recommendation, the atrio-caval junction may not be the best destination since a CVC tip so placed would be well seated within the pericardial sac, a vulnerable position concerning cardiac tamponade. This notion is based on anatomical data showing that the pericardial reflection extends for three cen timeters on the medial side of the SVC, which approximately corresponds to the mid-point of the SVC[7],[20]. One study involving embalmed cadavers reported the carina was situated at a mean distance of four millimeters above the pericardial reflection with an SE of one millimeter. In no single case was the carina seated lower than the level of the pericardial reflection[7]. These findings were corroborated by another anatomical study involving fresh cadavers[20].
Nonetheless, in vivo data showed that the carina did not assume the same relations with the pericardial sac or the midpoint of the SVC as those described in cadaveric studies. One radiological study conducted on live patients demonstrated that the upper limit of the pericardial reflection was approximately at the mid-point of the SVC and was consistently seen in the same computed tomography slices cutting through the takeoff of the right main bronchus. In contrast, the carina was situated lower than the pericardial reflection and the mid-point of the SVC by a mean (SD) of 0.7 (1.2) cm and 1.3 (0.9) cm, respectively. Therefore, the authors suggested using the right main bronchus takeoff as a more reliable in vivo surrogate landmark to minimize the risk of tamponade[8].
Figure 6. The percentage of CVC tips placed below the right main bronchus takeoff line as observed on postoperative chest radiographs for each of the four catheterization methods. Error bars represent the 95% confidence interval. The difference between the proposed surface landmarks rule and TEE did not exceed Sheskin’s minimum required difference (unstandardized difference [95% CI] = 14.3% [6.1%, 22.5%], Cohen’s dz [95% CI] = 0.22 [0.09, 0.35], P value = .004, Sheskin’s minimum required difference = 23.8%).
| Table 2. CVC length as estimated with TEE, the proposed surface landmarks rule, C-length, or Peres’ formula, and the CVC tip distance from the right main bronchus takeoff line on postoperative chest radiograph | |||||
|---|---|---|---|---|---|
| Variable | TEE | Proposed surface landmarks rule | C-length | Peres’ formula | P value |
| Estimated CVC length (mm) | 135.4 (132.8, 137.9) | 135.4 (133.8, 137.0) | 130.5 (127.0, 134.1)A | 163.8 (161.8, 165.8)B | < .0001* |
| CVC tip distance from the right main bronchus takeoff line (mm) t | -7.5 (-8.6, -6.5) | -7.5 (-10.0, -5.0) | -12.3 (-16.1, -8.6)A | 20.9 (17.6, 24.3)B | < .0001* |
| CVC tip sited below the right main bronchus takeoff line * | 0 (0.0% [0.0%, 5.2%]) | 10 (14.3% [7.9%, 24.3%])C | 20 (28.6% [19.4%, 40.0%])B | 64 (91.4% [82.5%, 96.0%])B | < .0001** |
Data are presented as mean (95% CI) or count (percentage [95% CI]). + Actual distance for TEE and expected distance for other methods. A minus (-) value refers to a location cephalad to the right main bronchus takeoff line, and a plus (+) value to a location caudad to the right main bronchus takeoff line; * Actual site for TEE and expected site for other methods; * Repeated-measures analysis of variance; ** Cochran Q test (Sheskin’s minimum significant difference = 23.8%);A P value = .003 versus TEE (Holm-Bonferroni-corrected); B P value < .0001 versus TEE (Holm-Bonferroni-corrected); C P value = .004 versus TEE ((Holm-Bonferroni-corrected); CVC = central venous catheter, TEE = transesophageal echocardiography.
A relevant issue that is worth discussing in this setting is the post-insertion migration of catheters, since there is data denoting that about 6% of initially well-placed CVCs undergo secondary migration or malposition[21]. Interestingly, a retrospective review of tunneled port catheters compared immediate post-insertion fluoroscopic screening images with chest radiographs taken within 48 hours of catheter placement. The authors reported that tip migration was observed in about 41% of catheters and that two-thirds of migrations occurred in women. Besides, they observed a statistically significant relation between tip migration and a higher body mass index (BMI) and female sex. They explained that women could be at higher risk for tip migration owing to the more massive breast tissue compared to males and concluded that allowance should be made for the possibility of migration, especially in women and
in those with higher BMI[22]. The implication of this is twofold: Firstly, any predictive rule should adjust for the possibility of catheter migration following proper placement, and secondly, rules designed to target more caudad destinations (e.g., lower SVC or atrio-caval junction) may not be a favorable choice if avoiding the occurrence of cardiac tamponade is a priority.
In the present trial, we demonstrated that the proposed surface landmarks rule was superior to the other two comparators, the C-length and Peres’ formula, both in terms of the magnitude of error in determining the required CVC length and in terms of the chance to have the CVC tip placed below the right main bronchus takeoff line. Of note, we demonstrated that Peres’ formula overestimated the needed depth of insertion by approximately 3 cm on average and could be associated with a frighteningly high chance for catheter tip placement below the right main bronchus takeoff that exceeded 90%. This is in concordance with a previous trial reporting that the vast majority (98.5%) of right IJV insertions guided with Peres’ formula were over-inserted[23]. Conversely, other investigators reported high accuracy of Peres’ formula for right IJV catheterization, but they defined different endpoints for the appropriateness of CVC tip position. For instance, Czepizak and coworkers[24] considered the placement as optimum if the tip was viewed above or in the distal SVC in anteroposterior chest radiographs and reported an accuracy of 90% for the formula. Contrary to Peres’ formula, the C-length underestimated the required depth of insertion by nearly 0.5 cm on average and was associated with a much lower chance of placing the CVC tip below the right main bronchus takeoff (about 29%) in the present study. This agrees with a previous series, which found that about 87% of the CVC tips inserted with the C-length guidance were placed in the upper SVC and only 13% in the lower SVC[9].
Although the precision of the C-length differed slightly from that of the proposed surface landmarks rule (SEest: 14.0 mm vs. 8.4 mm), the difference was more pronounced in the case of Peres’ formula (SEest: 30.7 mm). Deductively, the clinical implications of these differences could be relevant considering the observed radiographic findings, as approximately 30% of patients catheterized with guidance of the C-length method could have their CVC tip placed caudad to the right main bronchus takeoff line (i.e., within the pericardial reflection). For Peres’ formula, this probability could even be much higher, exceeding 90%. It is noteworthy that although the proposed surface landmarks rule was associated with a lower vulnerability in the order of 14% for this occurrence, given an estimated 95% CI of 7.9% to 24.3%, it is quite probable that a considerable proportion of catheterizations guided with the proposed rule could eventually end up with the CVC tip dwelling below the pericardial reflection. It need not be overstressed, therefore, that clinical vigilance should be exercised with whatever rule is employed to guide central venous catheterization.
Several methods and predictive rules, whether guided by topographical or radiological landmarks, have been proposed to help determine the ideal CVC insertion depth[3],[9],[23],[25]-[28], but none has been established as a benchmark. Intuitively, it cannot be anticipated that a uniform rule would be ubiquitously adopted by all practitioners, given the diversity in the clinical purpose for inserting a CVC and the variation in the metrics defining what is considered the ideal position of a CVC tip. For perioperative purposes, a CVC is customarily inserted for a relatively unprotracted duration, usually in the order of a few days, to administer medications and/or monitor right-sided filling pressure. In such a setting, the provision of a reliable method for verifying the location of the CVC tip, such as TEE, would not be feasible given the imposed costs, required expertise, and the ethical issues of subjecting patients to unnecessary interventions. Alternatively, performing intraoperative chest roentgenography could not be suggested as a routine practice for this purpose. The present trial, therefore, attempted to fill a relevant gap by seeking to develop a simple prediction rule based on the known surface projections of the great veins and the pericardial anatomy that are easily identified by clinicians. For this purpose, we determined the optimum CVC length required to place the tip at the target site (i.e., mid-SVC) with TEE, which we considered as our gold standard test given its demonstrated accuracy[29]. Besides, we evaluated the accuracy of the proposed rule for guiding placement of the CVC tip outside the pericardial reflection using a simple radiological landmark (i.e., right main bronchus takeoff) that has been demonstrated to be a better alternative to the carinal line[8].
On the other hand, the present study has limitations. Firstly, the accuracy of the suggested surface landmarks-based rule has been evaluated on a rather small cohort from which it was derived. A known shortcoming in predictive models is that they best fit the training sample from which they are developed[30]. Although we subjected the rule to internal validation using k-fold cross-validation, the rule has yet to undergo external validation on other separate samples that are adequately sized. Another limitation is that the prediction rule is meant to be applied to adult subjects undergoing right IJV cannulation utilizing a middle approach. The present results cannot, therefore, be generalized to other patient populations or other approaches for central venous cannulation.
In the context of limitations, it is also noteworthy to discuss the shortcomings of portable chest X-ray systems. The posteroanterior (PA) projection is considered the gold standard for chest radiography owing to its superior image quality and accurate cardiac size estimation; however, it is generally limited to ambulatory patients who can stand or sit fully upright at a radiographic unit. The reason is that a true PA view requires the X-ray beam to pass through the T7 level in a posterior-to-anterior projection at right angles to the image detector, which is placed against the patient’s anterior chest wall. The minimum source-to-image distance required to reduce the magnification of thoracic structures, especially the heart, and enhance image sharpness is 180 cm, which is usually unfeasible in a bedside setting[31]. For portable systems, however, the quality of the regularly performed AP projection could be significantly enhanced if patients were propped up in a 45to 60-degree head-up (Fowler’s) position, which is usually feasible, and sometimes the preferred position for nursing patients in the ICU. In contradistinction to the supine decubitus, this position limits cardiac magnification and enhances the accuracy of mediastinal assessment[32].
Another limitation is the effect of the body position and respiratory phase on the CVC tip position relative to the tracheobronchial tree. In this regard, there is evidence that the tip moves an average of 9 mm more cephalad to the carina during inspiration[33] or when changing from the supine to the upright position[34]. A peculiar, though unavoidable, problem akin to post-cardiac surgery radiographs is the potential to conceal mediastinal structures by wires, drains, or monitoring electrodes[35]. While it is possible that inter-rater variability could impact judgements, there is data denoting excellent inter-observer agreement for the determination of CVC tip on portable X-ray systems[36].
To minimize the effect of these confounding factors, we have standardized the portable imaging technique, addressing possible modifiable factors to enhance the reliability of radiographic images, which were examined by a single rater with adequate experience to reduce inter-rater variability.
Conclusion
The proposed surface landmarks-based rule presents a simple method to guide the determination of the CVC depth that is required to have the catheter tip placed at the mid-SVC in adults undergoing catheterization of the right IJV via the middle approach. The rule was associated with a smaller error and lower chance of intra-pericardial placement than both the C-length and Peres’ formula, and demonstrated better agreement with TEE than either method, but needs further validation on larger cohorts.
Statements and Declarations
Competing Interests: The authors have no relevant financial or non-financial interests to disclose.
Author Contributions:
All authors whose names are listed on this manuscript:
Ethics Approval: This study was performed following the principles of the Declaration of Helsinki. Approval was granted by the Research Ethics Committee of Ain Shams University Faculty of Medicine (identification number, FMASU MS 301/2019).
Consent to Participate: Written informed consent was obtained from all individual participants included in the study.
Referencias
1. Timsit JF. What is the best site for central venous catheter insertion in critically ill patients? Crit Care. 2003 Dec;7(6):397-9. https://doi.org/10.1186/cc2179 PMID:14624670
2. Fletcher SJ, Bodenham AR. Safe placement of central venous catheters: where should the tip of the catheter lie? Br J Anaesth. 2000 Aug;85(2):188-91. https://doi.Org/10.1093/bja/85.2.188 PMID:10992821
1. Peres PW. Positioning central venous catheters—a prospective survey. Anaesth Intensive Care. 1990 Nov;18(4):536-9. https://doi.org/10.1177/0310057X9001800422 PMID:2268022
2. Sheep RE, Guiney WB Jr. Fatal cardiac tamponade. Occurrence with other complications after left internal jugular vein catheterization. JAMA. 1982 Oct;248(13):1632-5. https://doi.org/10.1001/jama.1982.03330130080034 PMID:7109187
3. Collier PE, Ryan JJ, Diamond DL. Cardiac tamponade from central venous catheters. Report of a case and review of the English literature. Angiology. 1984 Sep;35(9):595-600. https://doi.org/10.1177/000331978403500909 PMID:6486523
4. Scott WL. Central venous catheters. An overview of Food and Drug Administration activities. Surg Oncol Clin N Am. 1995 Jul;4(3):377-93. https://doi.org/10.1016/S1055-3207(18)30433-2 PMID:7552783
5. Schuster M, Nave H, Piepenbrock S, Pabst R, Panning B. The carina as a landmark in central venous catheter placement. Br J Anaesth. 2000 Aug;85(2):192-4. https://doi.org/10.1093/bja/85.2.192 PMID:10992822
6. Caruso LJ, Gravenstein N, Layon AJ, Peters K, Gabrielli A. A better landmark for positioning a central venous catheter. J Clin Monit Comput. 2002 Aug;17(6):331-4. https://doi.org/10.1023/A:1024286119090 PMID:12885176
7. Lee JB, Lee YM. Pre-measured length using landmarks on posteroanterior chest radiographs for placement of the tip of a central venous catheter in the superior vena cava. J Int Med Res. 2010;38(1):134-41. https://doi.org/10.1177/147323001003800115 PMID:20233522
8. Cunningham DJ. Cunningham’s Textbook of Anatomy. 5th ed. New York: William Wood and Company; 1918. p. 1386.
9. Hamilton DF, Ghert M, Simpson AH. Interpreting regression models in clinical outcome studies. Bone Joint Res. 2015 Sep;4(9):152-3. https://doi.org/10.1302/2046-3758.49.2000571 PMID:26392591
10. McKinney W. Data structures for statistical computing in Python. In: van der Walt S, Millman J, eds. Proceedings of the 9th Python in Science Conference; June 28-July 3, 2010; Austin, TX. 2010:56-61. https://doi.org/10.25080/Majora-92bf1922-00a
11. Pedregosa F, Varoquaux G, Gramfort A, et al. Scikit-learn: machine learning in Python. J Mach Learn Res. 2011;12:2825-30. https://doi.org/10.5555/1953048.2078195
12. Python Software Foundation. Python 3.12.0 [computer program]. Wilmington, DE: Python Software Foundation; 2023. Accessed April 1, 2025. https://www.python.org
13. Altman DG, Bland JM. Measurement in medicine: the analysis of method comparison studies. Statistician. 1983;32(3):307-17. https://doi.org/10.2307/2987937
14. Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986 Feb;1(8476):307-10. https://doi.org/10.1016/S0140-6736(86)90837-8 PMID:2868172
15. Collier PE, Blocker SH, Graff DM, Doyle P. Cardiac tamponade from central venous catheters. Am J Surg. 1998 Aug;176(2):212- 4. https://doi.org/10.1016/S0002-9610(98)00171-8 PMID:9737635
16. Ballard DH, Samra NS, Gifford KM, Roller R, Wolfe BM, Owings JT. Distance of the internal central venous catheter tip from the right atrium is positively correlated with central venous thrombosis. Emerg Radiol. 2016 Jun;23(3):269-73. https://doi.org/10.1007/s10140-016-1393-2 PMID:27112774
17. Debourdeau P, Farge D, Beckers M, Baglin C, Bauersachs RM, Brenner B, et al. International clinical practice guidelines for the treatment and prophylaxis of thrombosis associated with central venous catheters in patients with cancer. J Thromb Haemost. 2013 Jan;11(1):71-80. https://doi.org/10.1111/jth.12071 PMID:23217208
18. Albrecht K, Nave H, Breitmeier D, Panning B, Tröger HD. Applied anatomy of the superior vena cava-the carina as a landmark to guide central venous catheter placement. Br J Anaesth. 2004 Jan;92(1):75-7. https://doi.org/10.1093/bja/aeh013 PMID:14665557
19. Vazquez RM, Brodski EG. Primary and secondary malposition of silicone central venous catheters. Acta Anaesthesiol Scand Suppl. 1985;81 s81:22-6. https://doi.org/10.1111/j.1399-6576.1985.tb02318.x PMID:3936326
20. Ryan D, Torreggiani W. Re: imaging and management of complications of central venous catheters. Clin Radiol. 2013 Nov;68(11):1181. https://doi.org/10.1016/j.crad.2013.06.015 PMID:23932677
21. Chopra S, Luthra N, Kathuria S, Saggar K, Gupta S, Singh T. Optimal positioning of right internal jugular venous catheter: A randomised study comparing modified Peres’ height formula and distance between insertion point and right third intercostal space. Indian J Anaesth. 2022 Aug;66(8):585-90. https://doi.org/10.4103/ija.ija_879_21 PMID:36274797
22. Czepizak CA, O’Callaghan JM, Venus B. Evaluation of formulas for optimal positioning of central venous catheters. Chest. 1995 Jun;107(6):1662-4. https://doi.org/10.1378/chest.107.6.1662 PMID:7781364
25. Ahn JH, Kim IS, Yang JH, Lee IG, Seo DH, Kim SP. Transoesoph- ageal echocardiographic evaluation of central venous catheter positioning using Peres’ formula or a radiological landmark-based approach: a prospective randomized single-centre study. Br J Anaesth. 2017 Feb;118(2):215-22. https://doi.org/10.1093/bja/aew430 PMID:28100525
26. Samerchua A, Lapisatepun P, Srichairatanakool P, et al. Evaluation of external and radiological landmark methods for optimizing ultrasound-guided right internal jugular venous catheterization depth in cardiac surgery. Sci Rep. 2025;15(1):1429. Published 2025 Jan 9. https://doi.org/10.1038/s41598-025- 85906-2
27. Manudeep AR, Manjula BP, Dinesh Kumar US. Comparison of Peres’ Formula and Radiological Landmark Formula for Optimal Depth of Insertion of Right Internal Jugular Venous Catheters. Indian J Crit Care Med. 2020 Jul;24(7):527-30. https://doi.org/10.5005/jp-journals-10071-23478 PMID:32963434
28. Ryu HG, Bahk JH, Kim JT, Lee JH. Bedside prediction of the central venous catheter insertion depth. Br J Anaesth. 2007 Feb;98(2):225-7. https://doi.org/10.1093/bja/ael339 PMID:17210734
29. Zhang M, Liu HL, Li WH, Li MZ. The value of transthoracic echocardiography and chest X-ray in locating the tip of central venous catheter in dialysis patients: a comparative study with computed tomography imaging. Ren Fail. 2023;45(2):2290179. https://doi.org/10.1080/0886022X.2023.2290179 PMID:38059492
10. Wasson JH, Sox HC, Neff RK, Goldman L. Clinical prediction rules. Applications and methodological standards. N Engl J Med. 1985 Sep;313(13):793-9. https://doi.org/10.1056/NEJM198509263131306 PMID:3897864
30. Johnson N, Brown K, Martensen KM. Chest. In: Lampignano JP, Kendrick LE, editors. Bontrager’s Textbook of Radiographic Positioning and Related Anatomy. 9th ed. St. Louis: Elsevier; 2018. pp. 71-104. ISBN: 978-0-323-39966-1.
31. Broder J. Imaging the chest: The chest radiograph. In: Broder J, editor. Diagnostic Imaging for the Emergency Physician. 1st ed. Philadelphia (PA): Elsevier-Saunders; 2011. pp. 185-296. https://doi.org/10.1016/B978-1-4160-6113-7.10005-5
32. Pan PP, Engstrom BI, Lungren MP, Seaman DM, Lessne ML, Kim CY. Impact of phase of respiration on central venous catheter tip position. J Vasc Access. 2013;14(4):383-7. https://doi.org/10.5301/jva.5000135 PMID:23599138
33. Nazarian GK, Bjarnason H, Dietz CA Jr, Bernadas CA, Hunter DW. Changes in tunneled catheter tip position when a patient is upright. J Vasc Interv Radiol. 1997;8(3):437-41. https://doi.org/10.1016/S1051-0443(97)70585-2 PMID:9152918
34. Chauhan A, Mujalde HS, Gupta M, Rathore DS. Comparative evaluation of central venous catheter tip positioning between post-procedure chest radiograph and intra-operative transesophageal echocardiography in cardiac surgery patients. Ann Card Anaesth. 2020;23(1):58-63. https://doi.org/10.4103/aca.ACA_147_1
35. Zhang X, Jia D, Ke N, Liu C, Fu L, Hu X. Excellent inter-observer agreement between radiologist and nurse: tracheal carina-based identification of peripherally inserted central catheter tip position. J Vasc Access. 2018 Jan;19(1):28-33. https://doi.org/10.5301/jva.5000799 PMID:29027181

 ORCID
ORCID