Miguel Andrés Bedoya López1, Fabricio Andrés Lasso Andrade3, José Hugo Arias Botero2, Catalina Moreno-Quijano2, María Camila Jaramillo Monsalve2, Susana Alzate Arguelles2, Andrés Yepes2
Recibido: 2025-08-24
Aceptado: 2025-09-28
©2026 El(los) Autor(es) – Esta publicación es Órgano oficial de la Sociedad de Anestesiología de Chile
Revista Chilena de Anestesia Vol. 55 Núm. 2 |https://doi.org/10.25237/revchilanestv55n2-10
PDF
Efectividad de la estimulación medular en el dolor crónico refractario: Un estudio de cohorte ambispectivo
Abstract
Introduction: Chronic pain is a public health issue with an estimated global prevalence ranging from 20.8% to 51.3%, leading to impaired quality of life. Spinal cord stimulation (SCS) has emerged as an effective therapy for pain syndromes refractory to medical management. The aim of this study was to evaluate the analgesic effectiveness and reduction in medication use following SCS in patients with refractory chronic pain. Methods: We conducted an observational, longitudinal, ambispective, and analytical study in patients aged > 18 years with CRPS, neuropathic pain, persistent spinal pain syndrome type 2, phantom limb pain, or chronic radicular pain who underwent SCS between May 2016 and August 2024. Pain intensity was assessed using the Numeric Rating Scale (NRS) and analgesic use before implantation, and at 3 and 6 months post-implantation. Responders were defined as patients achieving a > 50% reduction in NRS. Secondary outcomes included reduction in the use of opioids, NSAIDs, and neuromodulators. Results: A total of 49 patients were included; 51% were responders. At 6 months, responders showed a significant reduction in pain compared with non-responders (NRS 2.8 ± 1.32 vs. 6.42 ± 1.82; p < 0.001). SCS led to a significant decrease in the use of neuromodulators (98% to 67.3%), opioids (87.8% to 42.9%), and NSAIDs (42.9% to 6.1%), all with p < 0.001 at 6 months. The NNT was 3.3 for neuromodulators, 2.2 for opioids, and 2.7 for NSAIDs. Conclusion: SCS is an effective intervention that significantly reduces pain intensity and analgesic consumption within a 6-month period.
Resumen
Introduction: Chronic pain is a public health issue with an estimated global prevalence ranging from 20.8% to 51.3%, leading to impaired quality of life. Spinal cord stimulation (SCS) has emerged as an effective therapy for pain syndromes refractory to medical management. The aim of this study was to evaluate the analgesic effectiveness and reduction in medication use following SCS in patients with refractory chronic pain. Methods: We conducted an observational, longitudinal, ambispective, and analytical study in patients aged > 18 years with CRPS, neuropathic pain, persistent spinal pain syndrome type 2, phantom limb pain, or chronic radicular pain who underwent SCS between May 2016 and August 2024. Pain intensity was assessed using the Numeric Rating Scale (NRS) and analgesic use before implantation, and at 3 and 6 months post-implantation. Responders were defined as patients achieving a > 50% reduction in NRS. Secondary outcomes included reduction in the use of opioids, NSAIDs, and neuromodulators. Results: A total of 49 patients were included; 51% were responders. At 6 months, responders showed a significant reduction in pain compared with non-responders (NRS 2.8 ± 1.32 vs. 6.42 ± 1.82; p < 0.001). SCS led to a significant decrease in the use of neuromodulators (98% to 67.3%), opioids (87.8% to 42.9%), and NSAIDs (42.9% to 6.1%), all with p < 0.001 at 6 months. The NNT was 3.3 for neuromodulators, 2.2 for opioids, and 2.7 for NSAIDs. Conclusion: SCS is an effective intervention that significantly reduces pain intensity and analgesic consumption within a 6-month period.
Introduction
Chronic pain is a public health problem, with an estimated global prevalence ranging from 20.8%[1] to 51.3%[2] in the adult population. It is associated with functional impairment, high healthcare resource utilization, and reduced quality of life[3]. Complex regional pain syndrome (CRPS)[4] and persistent spinal pain syndrome type 2 (PSPS-T2) [5] continue to pose therapeutic challenges due to their limited response to conventional pharmacological treatment. Spinal cord stimulation (SCS) has become a consolidated therapeutic alternative for these syndromes, with growing evidence supporting its analgesic efficacy, reflected in reductions in the use of opioids, nonsteroidal anti-inflammatory drugs (NSAIDs), and neuromodulators[6].
The mechanism of action of SCS is explained by the stimulation of large-diameter afferent fibers in the dorsal columns of the spinal cord, which interferes with nociceptive transmission, based on the gate control theory proposed by Melzack and Wall[7]. However, experimental studies have demonstrated that its action is not limited to the spinal level but also involves supraspinal mechanisms[8], mediated by inhibitory factors such as gamma-aminobutyric acid (GABA) and other systems such as the cholinergic pathway[9], which inhibit wide dynamic range neurons through descending inhibitory mechanisms that may persist even after stimulation is turned off[10]. This has prompted investigations into its effectiveness across various chronic pain syndromes, although results remain inconclusive for many of them.
The aim of the present study was to evaluate the analgesic effectiveness of SCS in patients with different types of chronic pain refractory to medical management, treated at a referral center.
Methods
This study was approved by the Research Ethics Committee of the Colombian Pain Institute (INCODOL), as documented in the institutional minutes dated June 9, 2023. We conducted an observational, longitudinal, ambispective, and analytical study aimed at assessing the analgesic effectiveness of SCS in patients with chronic pain treated at INCODOL, in Medellín, Colombia. Patients who underwent SCS between May 2016 and August 2024 were included, with clinical follow-up up to six months after neurostimulator implantation.
The study population consisted of patients aged 18 years or older, with a clinical history of CRPS, neuropathic pain, PSPS-T2, phantom limb pain, or chronic radicular pain, with NRS > 7 and refractory to medical management. All patients underwent implantation of a dorsal column neurostimulator (Medtronic, Minneapolis, MN, USA) and had a clinical record documenting at least six months of post-procedural follow-up.
Collected variables included sociodemographic characteristics (age, sex, weight, height, and body mass index), clinical variables (diagnosis, pain location, medication use before and after the procedure, trial stimulation, physical therapy), and outcome variables (mean reduction in pain intensity on the Numeric Rating Scale, NRS) assessed before implantation and at 3 and 6 months post-implantation. The primary outcome was defined as a > 50% reduction in NRS score compared to baseline[11], measured at 3 and 6 months after implantation. Secondary outcomes included reductions in opioid, NSAID, and neuromodulator use after SCS implantation.
Data were collected using a Microsoft Excel® template by four investigators, who extracted the information directly from clinical records. Statistical analysis included descriptive statistics for quantitative variables expressed as measures of central tendency and dispersion (mean or median, standard deviation or interquartile range), according to variable distribution. Categorical variables were reported as absolute and relative frequencies. Normality was assessed using the Shapiro-Wilk test[12].
Patients were classified as responders or non-responders based on the primary outcome. Clinical and treatment characteristics were compared between groups. For categorical variables, chi-square or Fisher’s exact test was used, while for continuous variables, Student’s t-test was applied in cases of normal distribution or the Mann-Whitney U test[13] otherwise. To evaluate changes in NRS scores across the three measurement points (baseline, 3 months, and 6 months), Friedman’s non-parametric test[14] was applied, followed by paired Wilcoxon tests with Bonferroni correction for multiple comparisons. The number needed to treat (NNT) was calculated as the inverse of the absolute risk reduction in analgesic consumption.
Statistical analysis was performed using R language via RStudio® version 4.3.1, with statistical significance set at p < 0.05.
Results
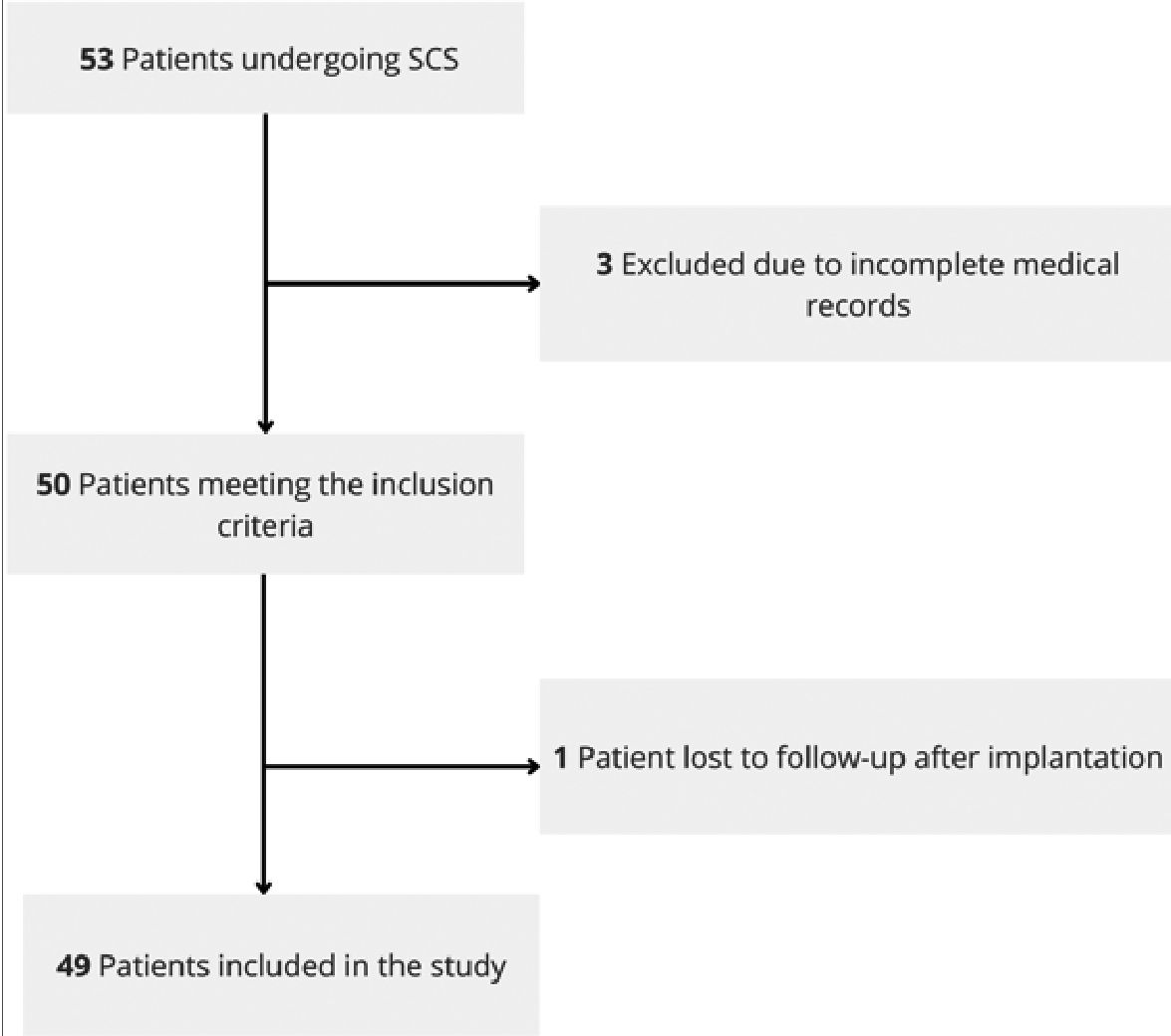
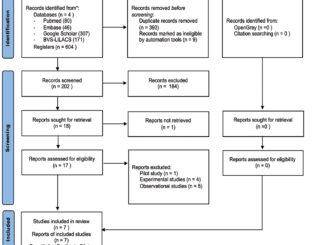
A total of 53 patients were screened for inclusion; after applying the eligibility criteria, 49 patients who underwent dorsal column stimulation for refractory chronic pain were included (Figure 1). Of these, 25 patients (51%) were classified as responders and 24 (49%) as non-responders. Most patients were male (61.2%), with a mean age of 46.9 ± 12.83 years. A significantly higher proportion of non-responders were not receiving any medication compared with responders (p = 0.048) (Table 1).
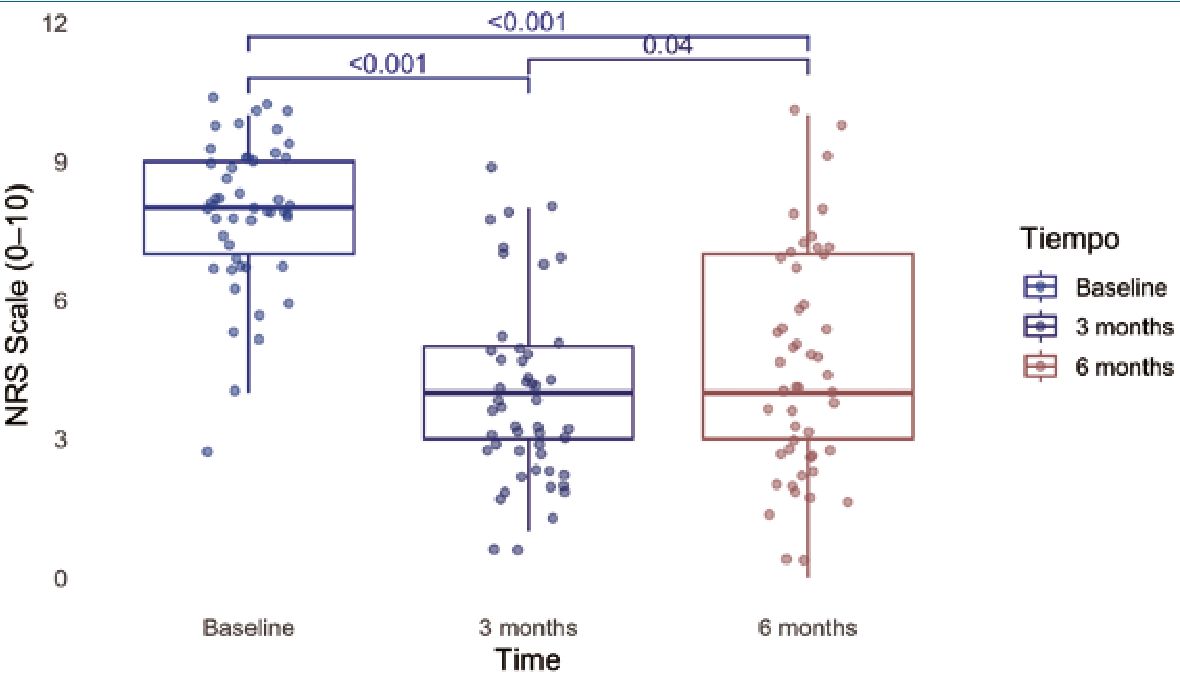
At baseline, mean NRS was 7.9 (SD ± 1.56). Responders had higher pain scores than non-responders (p = 0.044). This difference disappeared at 3 months post-implant (p = 0.143); however, at 6 months, the difference became significant again, with mean NRS 2.8 (SD ± 1.32) in responders vs. 6.42 (SD ± 1.82) in non-responders (p < 0.001). The clinical impact of SCS at 6 months was reflected in NNT values of 3.3 for neuromodulators, 2.2 for opioids, and 2.7 for NSAIDs (Table 2).
Figure 1. CONSORT diagram of patient inclusion, exclusion, and follow-up.
Figure 2. Comparison of NRS (0-10) before and after spinal cord stimulation across all patients at baseline, 3 months, and 6 months. Friedman test followed by pairwise Wilcoxon comparisons with Bonferroni correction was used.
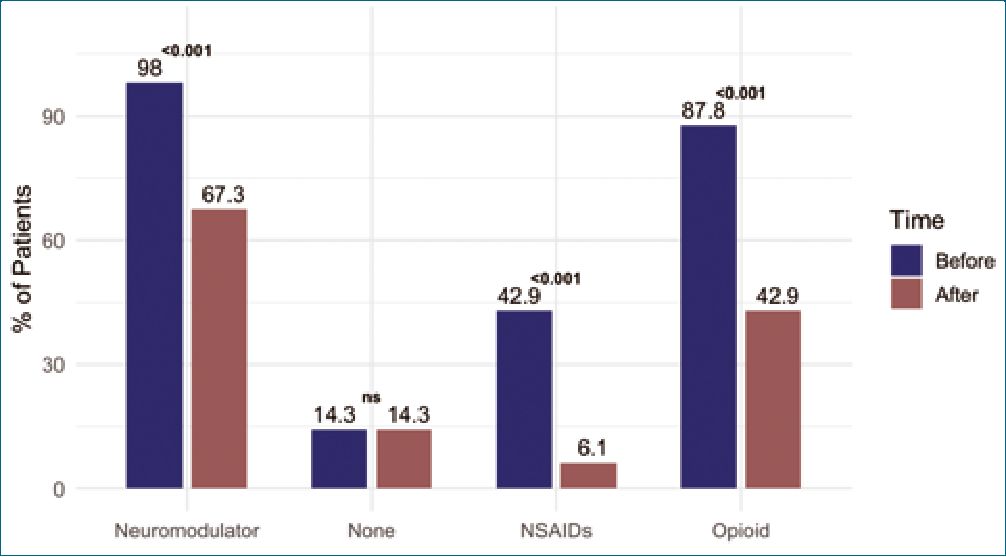
There was a significant reduction in pain over time in the overall cohort, reaching significance at both 3 and 6 months (Figure 2). This reduction was associated with decreased neuromodulator use, with greater improvement among responders (52%) compared to non-responders (83.3%) (p = 0.04). Overall, there was a significant reduction in neuromodulator use (98% vs. 67.3%, p < 0.001), opioids (87.8% vs. 42.9%, p < 0.001), and NSAIDs (42.9% vs. 6.1%, p < 0.001) after SCS implantation. No additional significant benefit was observed in patients without prior analgesic use (p = 0.19) (Figure 3).
Discussion
This ambispective cohort shows that SCS is an effective strategy for managing patients with refractory chronic pain. A total of 51% of patients achieved a clinically meaningful improvement in pain scores (> 50% NRS reduction), a success rate comparable to that reported for persistent spinal pain syndrome type 2 (PSPS-2) and diabetic neuropathy, which show success rates of 50%-80%[10],[15]. Pain reduction was sustained up to 6 months (p < 0.001), though longer follow-up is needed to evaluate long-term durability.
The limited efficacy of SCS over time is likely multifactorial. Tolerance is the main explanation for diminishing analgesic benefit[16]. One proposed mechanism is localized macroscopic fibrosis around the electrode tip, interfering with electrical current and ascending nociceptive pathways. Advances such as high-frequency and burst stimulation may help overcome tolerance[17]. Our study did not include patients with these newer modalities, which may account for limited long-term benefit in pain and medication reduction.
| Table 1. Demographic and clinical characteristics of patients | ||||
|---|---|---|---|---|
| Variable | Total sample n = 49 (%) | Responders n = 25 (%) | Non-responders n = 24 (%) | p-value |
| Gender | ||||
| Female | 19 (38.8) | 11 (44) | 8 (33.3) | 0.63* |
| Male | 30 (61.2) | 14 (56) | 16 (66.7) | |
| Age (mean ± SD) | 46.9 ± 12.83 | 46.8 ± 12.17 | 47 ± 13.74 | 0.94* |
| Weight (mean ± SD) | 72.95 ± 12.94 | 73.34 ± 14.46 | 72.54 ± 11.4 | 0.83* |
| BMI (mean ± SD) | 26.45 ± 4.35 | 26.39 ± 5.1 | 26.51 ± 3.5 | 0.46§ |
| Baseline NRS | ||||
| Mean ± SD | 7.9 ± 1.56 | 8.4 ± 1.12 | 7.38 ± 1.79 | 0.044§ |
| Median (IQR) | 8(7-9) | 8(8-9) | 8 (78) | |
| Diagnosis | ||||
| CRPS | 5 (10.2) | 4 (16) | 1 (4.2) | |
| Peripheral neuropathic pain | 14 (28.6) | 8 (32) | 6 (25) | |
| Persistent spinal pain syndrome type 2 | 14 (28.6) | 7 (28) | 7 (29.2) | 0.21 + |
| Phantom limb pain | 4 (8.2) | 3 (12) | 1 (4.2) | |
| Chronic radicular pain | 12 (24.5) | 3 (12) | 9 (37.5) | |
| Electrode location | ||||
| Cervical | 14 (28.6) | 6 (24) | 8 (33.3) | |
| Thoracic | 28 (57.1) | 17 (68) | 11 (45.8) | 0.24+ |
| Lumbar | 7 (14.3) | 2 (8) | 5 (20.8) | |
| Opioids prior to SCS | 43 (87.8) | 24 (96) | 19 (79.2) | 0.09+ |
| NSAIDs prior to SCS | 21 (42.9) | 10 (40) | 11 (45.8) | 0.902* |
| No analgesics prior to SCS | 7 (14.3) | 1 (4) | 6 (25) | 0.048+ |
| Neuromodulators prior to SCS | 48 (98) | 25 (100) | 23 (95.8) | 0.49+ |
| Physical therapy | 26 (53.1) | 12 (48) | 14 (58.3) | 0.66* |
| Neurostimulation trial | 47 (95.9) | 25 (100) | 22 (91.7) | 0.23+ |
Comparison of clinical and therapeutic characteristics between responders and non-responders to spinal cord stimulation (SCS). Values are expressed as n (%) for categorical variables, mean ± SD for normally distributed continuous variables, and median (IQR Q1-Q3) for nonnormally distributed variables. *Chi-square test; tFisher’s exact test; tStudent’s t-test; §Mann-Whitney U test. SD: standard deviation; SCS: spinal cord stimulator; CRPS: complex regional pain syndrome; BMI: body mass index; NRS: Numerical Rating Scale; IQR: interquartile range; NSAID: nonsteroidal anti-inflammatory drug.
| Table 2. Outcomes after spinal cord stimulation implant | ||||
|---|---|---|---|---|
| Variable | Total sample n = 49 (%) | Responders n = 25 (%) | Non-responders n = 24 (%) | p-value |
| NRS at 3 months | ||||
| Mean ± SD | 3.94 ± 1.99 | 3.52 ± 1.66 | 4.38 ± 2.24 | 0.143* |
| Median (IQR) | 4 (3 – 5) | 3 (2 – 4) | 4 (3 – 5) | |
| NRS at 6 months | ||||
| Mean ± SD | 4.57 ± 2.41 | 2.8 ± 1.32 | 6.42 ± 1.82 | < 0.001* |
| Median (IQR) | 4 (3 – 7) | 3 (2 – 4) | 7 (5 – 7) | |
| Neuromodulators | 33 (67.3) | 13 (52) | 20 (83.3) | 0.04* |
| Opioids | 21 (42.9) | 10 (40) | 11 (45.8) | 0.9* |
| NSAIDs | 3 (6.1) | 2 (8) | 1 (4.2) | 0.99+ |
| No analgesics or neuromodulators | 7 (14.3) | 6 (24) | 1 (4.2) | 0.09+ |
Values presented for the total cohort, responders, and non-responders. *Chi-square test; tFisher’s exact test; tMann-Whitney U test. SD: standard deviation; SCS: spinal cord stimulator; NRS: Numerical Rating Scale; IQR: interquartile range; NSAID: nonsteroidal anti-inflammatory drug.
Figure 3. Comparison of the proportion of patients using each medication type before and 6 months after spinal cord stimulation. McNemar test was used for paired comparisons of pre- and post-implant medication use. NS: not significant.
A key goal of SCS is reducing medication use[18]. We observed significant and clinically relevant reductions in opioids (p < 0.001), NSAIDs (p < 0.001), and neuromodulators (p <0.001) within 6 months. NNT values of 2.2 for opioids, 2.7 for NSAIDs, and 3.3 for neuromodulators highlight the substantial pharmacological burden reduction[19]. Given the global public health crisis of opioid dependence, reducing reliance on medications with significant side effects and dependency risk may improve safety and quality of life in refractory chronic pain patients[20].
Our results reflect the diversity of real-world chronic pain populations. While most evidence focuses on CRPS[21] and PSPS-2[10], our findings suggest SCS may benefit other conditions such as radicular pain and phantom limb pain, though further studies are needed. Real-world data such as ours add value, often better reflecting clinical complexity than highly controlled RCTs[22]. This supports the external validity of current evidence in the Latin American context, with efficacy similar to that reported globally.
Limitations
This study has several limitations. First, only patients with >6 months follow-up were included, potentially biasing responder rates. However, only 7.5% of eligible patients were excluded, mainly due to incomplete records. Second, heterogeneity in diagnoses (CRPS, PSPS-2, radicular pain) reduces condition-specific conclusions, although it reflects real clinical practice. While SCS efficacy in PSPS-2 and CRPS is well established, its benefit in phantom limb pain remains preliminary[23]. Third, the absence of a control group (placebo or standard medical therapy) limits causal attribution. Fourth, follow-up was limited to 6 months, whereas other studies show declining efficacy beyond 3 years[24], leading to explantation[25]. Lastly, as a single-center study, findings may not generalize and could reflect center-specific expertise.
Future directions
Prospective, multicenter studies with longer follow-up are needed to confirm efficacy in heterogeneous populations, given potential attenuation of pain relief over time[25]. Further research should explore less-studied conditions such as phantom limb pain, where early results are promising. Incorporating patient-centered outcomes such as quality of life, functionality, and return to work will help evaluate the broader impact of SCS and refine patient selection for optimal benefit.
Conclusion
Spinal cord stimulation shows 51% effectiveness in refractory chronic pain patients, sustained for at least 6 months. This benefit is accompanied by significant reductions in neuromodulator, opioid, and NSAID use, underscoring its potential to reduce pharmacological burden and associated risks. Multicenter, long-term studies are required to confirm sustained efficacy and evaluate broader impacts on quality of life and functionality.
Authors’ contributions:
Referencias
1.Nahin RL, Feinberg T, Kapos FP, Terman GW. Estimated Rates of Incident and Persistent Chronic Pain Among US Adults, 2019-2020. JAMA Network Open [Internet]. 2023 May 16;6(5):e2313563. Available from: http://dx.doi.org/10.1001/jamanetworkopen.2023.13563
2.Fayaz A, Croft P, Langford RM, Donaldson LJ, Jones GT. Prevalence of chronic pain in the UK: a systematic review and meta-analysis of population studies. BMJ Open. 1 de junio de 2016;6(6):e010364. https://doi.org/10.1136/bmjop-en-2015-010364
3.Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. The Lancet. 15 de diciembre de 2012;380(9859):2163-96.
4.Ferraro MC, O’Connell NE, Sommer C, Goebel A, Bultitude JH, Cashin AG, et al. Complex regional pain syndrome: advances in epidemiology, pathophysiology, diagnosis, and treatment. Lancet Neurol. mayo de 2024;23(5):522-33. https://doi.org/10.1016/S1474-4422(24)00076-0
5.van de Minkelis J, Peene L, Cohen SP, Staats P, Al-Kaisy A, Van Boxem K, et al. 6. Persistent spinal pain syndrome type 2. Pain Pract. septiembre de 2024;24(7):919-36.
6.Kaye AD, Archer JR, Shah S, Johnson CD, Herron LR, Brouillette AE, et al. Spinal Cord Stimulation for Low Back Pain: A Systemat¬ic Review. Curr Pain Headache Rep. 2 de enero de 2025;29(1):2. https://doi.org/10.1007/s11916-024-01336-1
7.Melzack R, Wall PD. Pain mechanisms: a new theory. Science. 19 de noviembre de 1965;150(3699):971-9. https://doi.org/10.1126/ science.150.3699.971.
8.Caylor J, Reddy R, Yin S, Cui C, Huang M, Huang C, et al. Spinal cord stimulation in chronic pain: evidence and theory for mech¬anisms of action. Bioelectron Med. 28 de junio de 2019;5:12. https://doi.org/10.1186/s42234-019-0023-1
9.Song Z, Ultenius C, Meyerson BA, Linderoth B. Pain relief by spinal cord stimulation involves serotonergic mechanisms: an experimental study in a rat model of mononeuropathy. Pain. 15 de diciembre de 2009;147(1-3):241-8. https://doi.org/10.1016/j.pain.2009.09.020
10.North RB, Kidd DH, Petrucci L, Dorsi MJ. Spinal cord stimulation electrode design: a prospective, randomized, controlled trial comparing percutaneous with laminectomy electrodes: part II-clinical outcomes. Neurosurgery. noviembre de 2005;57(5):990-6; discussion 990-996.
11.Farrar JT, Young JP, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. PAIN. 1 de noviembre de 2001;94(2):149-58.
12.SHAPIRO SS, WILK MB. An analysis of variance test for normality (complete samples)+. Biometrika. 1 de diciembre de 1965;52(3- 4):591-611.
13.Mann HB, Whitney DR. On a Test of Whether one of Two Random Variables is Stochastically Larger than the Other. The Annals of Mathematical Statistics. marzo de 1947;18(1):50-60. https://doi.org/10.1214/aoms/1177730491
14.Friedman M. The Use of Ranks to Avoid the Assumption of Normality Implicit in the Analysis of Variance. Journal of the American Statistical Association. 1 de diciembre de 1937;32(200):675- 701. https://doi.org/10.1080/01621459.1937.10503522
15.de Vos CC, Meier K, Zaalberg PB, Nijhuis HJA, Duyvendak W, Vesper J, et al. Spinal cord stimulation in patients with painful diabetic neuropathy: a multicentre randomized clinical trial. Pain. noviembre de 2014;155(11):2426-31. https://doi.org/10.1016/j.pain.2014.08.031
16.Kumar K, Nath R, Wyant GM. Treatment of chronic pain by epidural spinal cord stimulation: a 10-year experience. J Neurosurg. septiembre de 1991;75(3):402-7.
17.Isagulyan E, Slavin K, Konovalov N, Dorochov E, Tomsky A, Dekopov A, et al. Spinal cord stimulation in chronic pain: technical advances. Korean J Pain. 1 de abril de 2020;33(2):99-107. https://doi.org/10.3344/kjp.2020.33.2.99
18.Yong RJ, Tran OV, McGovern AM, Patil PG, Gilligan CJ. LongTerm Reductions in Opioid Medication Use After Spinal Stimulation: A Claims Analysis Among Commercially-Insured Population. JPR. 17 de mayo de 2024;17:1773-84. https://doi.org/10.2147/JPR.S441195
19.Lo Bianco G, Al-Kaisy A, Natoli S, Abd-Elsayed A, Matis G, Papa A, et al. Neuromodulation in chronic pain management: addressing persistent doubts in spinal cord stimulation. Journal of Anesthesia, Analgesia and Critical Care. 6 de enero de 2025;5(1):3. https://doi.org/10.1186/s44158-024-00219-6
20.Dowell D, Compton WM, Giroir BP. Patient-Centered Reduction or Discontinuation of Long-term Opioid Analgesics. JAMA. 19 de noviembre de 2019;322(19):1855-6. https://doi.org/10.1001/jama.2019.16409
21.Schwarm FP, Stein M, Uhl E, Maxeiner H, Kolodziej MA. Spinal cord stimulation for the treatment of complex regional pain syndrome leads to improvement of quality of life, reduction of pain and psychological distress: a retrospective case series with 24 months follow up. Scand J Pain. 28 de abril de 2020;20(2):253-9. https://doi.org/10.1515/sjpain-2019-0081
22.Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpre¬tation and Impact of Real-World Clinical Data for the Practicing Clinician. Adv Ther. 2018 Nov;35(11):1763-74. https://doi.org/10.1007/s12325-018-0805-y PMID:30357570
23.Lee PM, So Y, Park JM, Park CM, Kim HK, Kim JH. Spinal Cauda Equina Stimulation for Alternative Location of Spinal Cord Stimu¬lation in Intractable Phantom Limb Pain Syndrome: A Case Report. The Korean Journal of Pain. 1 de abril de 2016;29(2):123-8. https://doi.org/10.3344/kjp.2016.29.2.123
24.Kemler MA, Vet HCW de, Barendse GAM, Wildenberg FAJM van den, Kleef M van. Spinal Cord Stimulation for Chronic Reflex Sympathetic Dystrophy – Five-Year Follow-up. New England Journal of Medicine. 1 de junio de 2006;354(22):2394-6.
25.Song IA, Lee JH, Han WK, Nahm FS. The actual duration of spinal cord stimulator use in patients with complex regional pain syndrome: a Korean nationwide cohort study. The Korean Journal of Pain. 1 de enero de 2025;38(1):51-7. https://doi.org/10.3344/kjp.24209

 ORCID
ORCID