Ahmed Abd Elnaiem Mohamed1, Rasha Ahmed Ali1, Ekram Abdullah Osman1
Recibido: 2025-06-01
Aceptado: 2025-09-21
©2026 El(los) Autor(es) – Esta publicación es Órgano oficial de la Sociedad de Anestesiología de Chile
Revista Chilena de Anestesia Vol. 55 Núm. 2 |https://doi.org/10.25237/revchilanestv55n2-11
PDF
Efecto de la infusión intraoperatoria de lidocaína sobre la inducción de la motilidad intestinal y la liberación de citocinas proinflamatorias inducida por la cirugía después de la cirugía abdominal: Un estudio clínico prospectivo, comparativo, aleatorizado, doble ciego y controlado
Abstract
Background: Lidocaine protects cells from inflammation by blocking the priming of neutrophils and therefore inhibiting the release of superoxide anions. Perioperative administration of lidocaine was significantly associated with attenuation of surgery-induced release of pro- inflammatory cytokines, e.g., IL-6 and IL-8, and/or decreased C-reactive protein levels. The aim of this work was investigating the ability of lidocaine local anesthetic on enhancing return of bowel motility after abdominal surgery. Methods: This prospective, randomized, double-blind controlled clinical study was carried out on 60 patients scheduled for elective intestinal surgery, body mass index (BMI) 18 to 30 kgm-2 and ASA I – II. They were randomly allocated into two equal groups: Group A: received general anesthesia and 1.5 mg/kg lidocaine at the induction of anesthesia followed by an infusion rate of 1.5 mg/kg/h for 4 h and Group B: general anesthesia and normal saline infusion (placebos). Results: Visual analogue scale was significantly lower at 2 h, 6 h, 12 h and 24 h in group A than group B (P value < 0.05). Interleukin-6 was significantly lower at 8 h after incision and 24 h after surgery in group A than group B (P < 0.001). Time of sounds, time of flatus and time of defecation were significantly lower in group A than group B (P value < 0.05). Conclusions: Intraoperative intravenous lidocaine infusion improved postoperative pain control, delayed the need for analgesia, and reduced inflammation, as shown by lower interleukin-6 levels. Additionally, it significantly accelerated bowel recovery, evidenced by earlier bowel sounds, flatus, and defecation, highlighting its potential to enhance postoperative recovery in elective intestinal surgery.
Resumen
Background: Lidocaine protects cells from inflammation by blocking the priming of neutrophils and therefore inhibiting the release of superoxide anions. Perioperative administration of lidocaine was significantly associated with attenuation of surgery-induced release of pro- inflammatory cytokines, e.g., IL-6 and IL-8, and/or decreased C-reactive protein levels. The aim of this work was investigating the ability of lidocaine local anesthetic on enhancing return of bowel motility after abdominal surgery. Methods: This prospective, randomized, double-blind controlled clinical study was carried out on 60 patients scheduled for elective intestinal surgery, body mass index (BMI) 18 to 30 kgm-2 and ASA I – II. They were randomly allocated into two equal groups: Group A: received general anesthesia and 1.5 mg/kg lidocaine at the induction of anesthesia followed by an infusion rate of 1.5 mg/kg/h for 4 h and Group B: general anesthesia and normal saline infusion (placebos). Results: Visual analogue scale was significantly lower at 2 h, 6 h, 12 h and 24 h in group A than group B (P value < 0.05). Interleukin-6 was significantly lower at 8 h after incision and 24 h after surgery in group A than group B (P < 0.001). Time of sounds, time of flatus and time of defecation were significantly lower in group A than group B (P value < 0.05). Conclusions: Intraoperative intravenous lidocaine infusion improved postoperative pain control, delayed the need for analgesia, and reduced inflammation, as shown by lower interleukin-6 levels. Additionally, it significantly accelerated bowel recovery, evidenced by earlier bowel sounds, flatus, and defecation, highlighting its potential to enhance postoperative recovery in elective intestinal surgery.
Introduction
Lidocaine is an amide local anaesthetic and an antiarrhythmic agent, first synthesized in 1942, and after approval for human use was launched in 1948 in Sweden. The first observations of post operative analgesic effects of perioperative intravenous lidocaine (IVL) were initially proposed in 1951 Subsequently many more enthusiastic reports followed[1].
In the late 1950s where IVL was demonstrated to have a postoperative analgesic effect without posing the risk of respiratory depression, reducing the occurrence of postoperative nausea and vomiting (PONV), and enhancing post-operative recovery[2].
IVL also potentiated the depth of anesthesia and led to a better tolerance of endotracheal intubation[3].
Around 40% of patients experience a delay in resumption of normal bowel function after colorectal surgery[4].
This delay leads to symptoms of nausea, vomiting, constipation, and abdominal distension, which then require unpleasant supportive interventions such as intravenous fluids and nasogastric tube insertion. ALLEGRO, A placebo-controlled randomized trial of IVL in accelerating Gastrointestinal Recovery after colorectal surgery, is the latest ongoing multicenter research study across the United Kingdom, investigating the use of IVL to improve recovery after colorectal surgery[5].
Evidence corroborated from various meta-analyses in the past have shown that perioperative lidocaine infusion at. Dose of 1.5 to 3 mg/kg/h consistently improved postoperative Visual Analogue Scale (VAS). Pain scores in patients undergoing either open or laparoscopic abdominal surgery[6].
However, it is important to note that the recommended dose of continuous lidocaine infusion should be no more than 1.5 mg/kg/h. Intraoperative and operative opioid requirements were also decreased with the use of lidocaine. In addition to reducing pain, another benefit is a reduction in the duration of postoperative ileus by an average of eight hours[7].
Lidocaine possesses an anti-inflammatory property, Lidocaine, as well as other amide local anaesthetic, inhibits leukocyte activation and adhesion to the site of injury in both in vitro and in vivo models[8]. Lidocaine protects cells from inflammation by blocking the priming of neutrophils and therefore inhibiting the release of superoxide anions[1].
Perioperative administration of lidocaine was significantly associated with attenuation of surgery-induced release of pro-inflammatory cytokines, e.g., IL-6 and IL-8, and/or decreased C-reactive protein (CRP) levels[9].
The aim of this work was investigating the ability of lidocaine local anesthetic on enhancing return of bowel motility after abdominal surgery.
Patients and Methods
This prospective, randomized, double-blind controlled clinical study was carried out on 60 patients scheduled for elective intestinal surgery, body mass index (BMI) 18 to 30 kg-m2 and ASA I – II, aged from 18 to 60 years old, both sexes. The study was done from October 2022 to August 2024 after approval from the Ethical Committee Assiut University, Assiut, Egypt (approval code: IRB 17101958) and registration of clinicaltrials. gov (ID: NCT05541640). An informed written consent was obtained from the patient or relatives of the patients.
The exclusion criteria were patients with preoperative gastrointestinal dysfunction, a history of drug abuse, or long-term opioid use and previous gastrointestinal surgery.
Randomization and blindness
It was done by computer-generated random tables in blocks of 6 numbers, and 1:1 allocation ratio; numbers were kept in well-sealed envelopes opened immediately before anesthesia induction by an anesthetist other than the anesthetist in charge or who is responsible for data collection.
Patients were randomly allocated into two equal groups: Group A: Received general anesthesia and 1.5 mg/kg lidocaine at the induction of anesthesia followed by an infusion rate of 1.5 mg/kg/h for 4 h. Group B: Received general anesthesia and normal saline infusion (placebos).
Preoperative preparation
The medical and surgical histories of the patients were taken, clinical examinations were performed, and routine laboratory investigations such as CBC, INR, coagulation studies, renal function, liver function, electrocardiography (ECG), and ECHO (on need) were performed.
Anesthesia induction
General anesthesia induced by propofol 1.5 mg/kg, titrated slowly till loss of eyelash reflex, cisatracurium 0.15 mg/kg, fentanyl 1 mic/kg, and lidocaine 1.5 mg/kg to facilitate stress-free endotracheal intubation. Anesthesia is maintained by isoflurane inhalation anesthesia, the cisatracurium maintenance dose of 0.03 mg/kg every 20 minutes, and FIO2 100%. Intraoperative analgesia was 10 mg of nalupuphine given immediately after intubation. At the end of surgery, the reversal of neuromuscular blockade was done by neostigmine 2.5 mg+1mg atropine, and extubation was done only after patients fulfill the criteria of extubation. Patients were monitored in PACU for at least 30 minutes and then transferred to a postoperative high dependency unit for 24 h postoperative.
Data collection
|Demographic data as age, sex, height, weight and ASA. Preoperative baseline vital sign parameters of heart rate (H.R), mean arterial pressure (M.A.P), and oxygen saturation (SPO2) were recorded. Intraoperative MAP, HR, and SpO2 % were monitored continuously throughout the operation and recorded every 5 minutes for the first 15 minutes and every 10 minutes until the end of the procedure. Surgery duration was defined as the time between the surgical incision and wound closure.
Postoperative data
Pain assessment by VAS score at 2 h, 6 h, 12 h and 24 h postoperative. First postoperative anal venting: patients were instructed to inform the nurse about the 1st flatus passage and the time of first defecation, a nurse in charge was informed to assess flatus passage and defecation by direct question to the patient every 2 hours. Intestinal sounds were evaluated every 4 h by the surgeon resident in charge. IL6 was measured preoperative, and 8 h and 24 h postoperative.
The time of the 1st analgesia request was recorded by the nurse in charge. When a patient first asked for analgesia, an anesthetist was informed to evaluate the pain score. Rescue analgesia was prescribed as paracetamol 1g if VAS < 4 and nalupuphine 6 to 8 mg shots if VAS > 4.
The primary outcome was the time of first postoperative anal venting. Anal venting is defined as the interval from the completion of surgical suturing until the patient’s first act of anal venting (First hearing of intestinal sounds, first passage of flatus, first defecation). The secondary outcomes were serum level of IL6 measured preoperative and 24 h postoperative. Time of the first analgesia. 24 h VAS score.
Sample size calculation
The sample size was calculated using G* Power analysis. Depending on the results of a previous study[10] the sample size was estimated to be able to detect a 20% difference in the meantime of return of the bowel function with an alpha level of 0.05, a beta level of 0.1 a minimum of 28 patients were required in each group to achieve 85% power. The sample was raised to include 30 patients/group for better and robust inferential statistics. Patients, data collectors, and analysts were blind to the study groups.
Statistical analysis
Statistical analysis was done by SPSS version 26 (IBM Inc., Chicago, IL, USA). Shapiro-Wilks test and histograms were used to evaluate the normality of the distribution of data. Quantitative parametric variables were presented as mean and standard deviation (SD) and compared between the two groups utilizing an unpaired Student’s T-test. Quantitative non-parametric data were presented as the median and interquartile range (IQR) and were analyzed by Mann Whitney test. Qualitative variables were presented as frequency and percentage (%) and were analyzed utilizing the Chi-square test or Fisher’s exact test when appropriate. A two-tailed P value < 0.05 was considered statistically significant.
Results
Seventy-three patients were assessed for eligibility, 9 patients did not meet the criteria, and 4 patients refused to participate in the study. The remaining patients were randomly allocated into two equal groups (30 patients in each). All allocated patients were followed up and analyzed statistically (Figure 1).
Age, sex, BMI, ASA and duration of surgery were insignificantly different between both groups (Table 1).
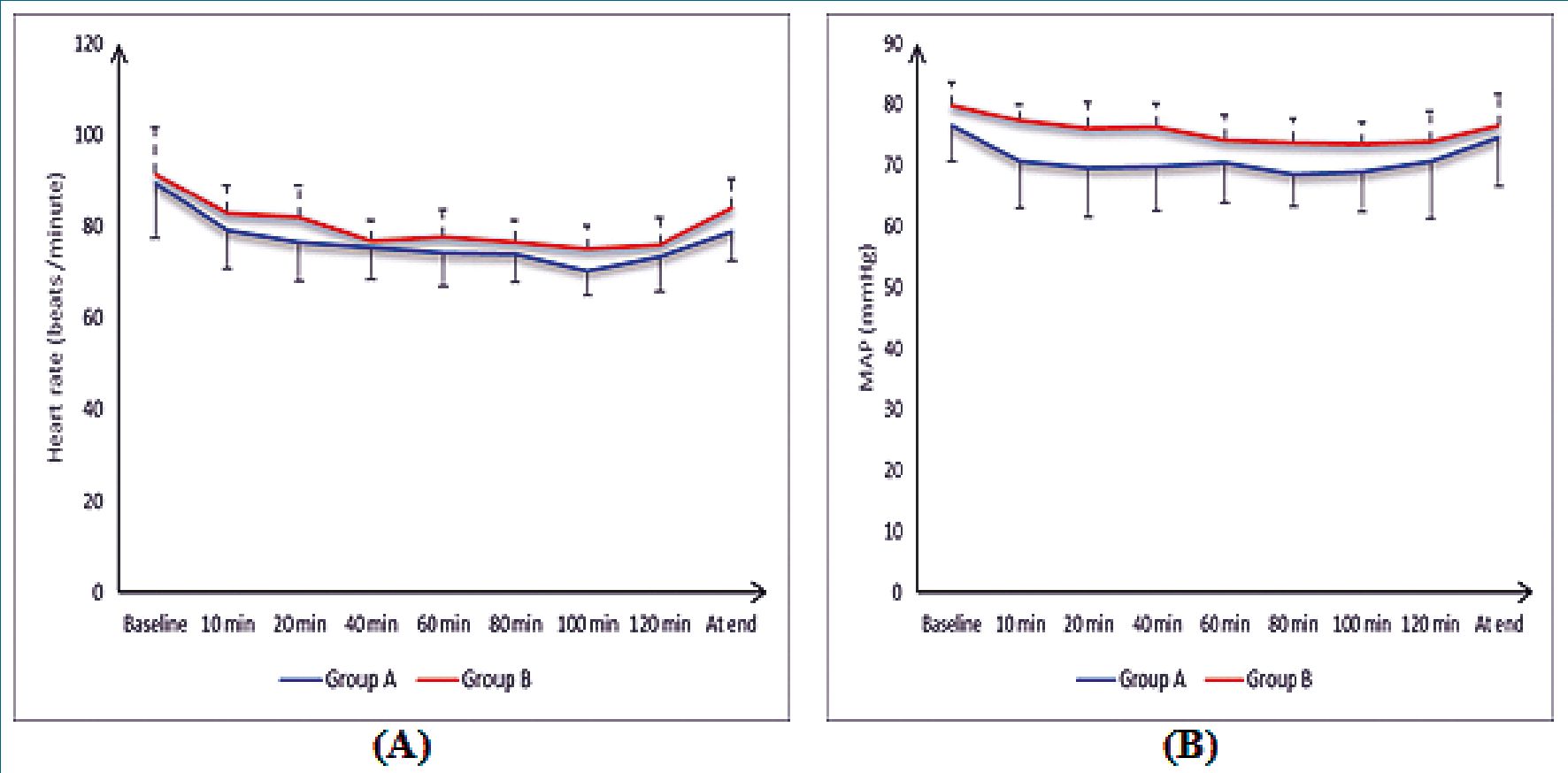
The heart rate was insignificantly different at baseline, 10 min, 40 min, 60 min, 80 min and 120 min between both groups. The heart rate was significantly lower at 20 min, 100 min and at the end in group A than group B (P < 0.05). The mean arterial blood pressure (MAP) was insignificantly different at 120 min and at end between both groups. The MAP was significantly lower at baseline, 10 min, 20 min, 40 min, 60 min, 80 min and at 100 min group A than group B (P < 0 .05) (Figure 2).
VAS was significantly lower at 2 h, 6 h, 12 h and 24 h in group A than group B (P value < 0.05) (Table 2).
Interleukin-6 was significantly lower at 8 h after incision and 24 hours after surgery in group A than group B (P < 0.001). Time of sounds, time of flatus and time of defecation were significantly lower in group A than group B (P value < 0.05) (Table 3).
Discussion
Abdominal surgery is the most common type of major surgery conducted in the world[11],[12]. These surgeries are often associated with postoperative pain, nausea, ileus, and extended hospital stays. Opioids have traditionally been the primary choice for perioperative pain management; however, their use is closely linked to postoperative ileus, particularly when daily dosing exceeds 2 mg of intravenous (IV) hydromorphone equivalents[13],[14].
Figure 1. CONSORT flowchart of the enrolled patients.
| Table 1. Demographic data and duration of surgery of the studied groups | ||||
|---|---|---|---|---|
| Group A (n = 30) | Group B (n = 30) | P | ||
| Age (years) | 45.37 ± 14.62 | 48.03 ± 10.16 | 0.4 | |
| Sex | Male | 17 (56.67%) | 19 (63.33%) | 0.5 |
| Female | 13 (43.33%) | 11 (36.67%) | ||
| BMI (kg/m2) | 27.4 ± 2.04 | 27.67 ± 1.69 | 0.6 | |
| ASA physical status | I | 17 (46.67%) | 13 (43.3%) | 0.4 |
| II | 13 (43.33%) | 17 (46.67%) | ||
Data are presented as mean ± SD or frequency (%); BMI: Body mass index.
In the current study, the intraoperative heart rate was insignificantly different at baseline, 10 min, 40 min, 60 min, 80 min and 120 min between both groups. The intraoperative heart rate was significantly lower at 20 min, 100 min and at the end in lidocaine group than placebo group. The intraoperative MAP was insignificantly different at 120 min and at end between both groups. The intraoperative MAP was significantly lower at baseline, 10 min, 20 min, 40 min, 60 min, 80 min and at 100 min lidocaine group than placebo group.
In agreement with our study, Sarakatsianou et al.[15], illustrated that lidocaine resulted in reduced intraoperative systolic, diastolic, and mean arterial pressure.
This was consistent with Hassan et al.[16], stated that the lidocaine group had a significantly lower MAP and heart rate compared to the control group.
However, Choi et al.[17], stated that the hemodynamic including heart rate and MAP were similar between lidocaine group and the control group. This disparity in results could be explained by the differences in the types of surgery performed, patients’ characteristics and study areas.
Figure 2. (A) Heart rate (B) MAP of the studied groups.
| Table 2. VAS, duration of anesthesia and time of 1st analgesia of the studied groups | |||
|---|---|---|---|
| Group A (n = 30) | Group B (n = 30) | P | |
| 2 h postoperative | 1 ± 0.3 | 1.5 ± 0.5 | 0.004* |
| 6 h postoperative | 1.7 ± 0.4 | 2.13 ± 0.6 | 0.002* |
| 12 h postoperative | 1.8 ± 0.3 | 2.5 ± 0.5 | < 0.001* |
| 24 h postoperative | 2.2 ± 0.4 | 2.7 ± 0.6 | 0.002* |
| Duration of anesthesia min | 164.47 ± 14.55 | 151.57 ± 11.46 | < 0.001* |
| Time of 1st analgesia (h) | 5 ± 0.8 | 2.2 ± 1 | < 0.001* |
| Data was presented as a mean (SD±). | ; VAS: Visual analog scale; *: | Significant as P value < 0.05. | |
Data was present as mean (±SD); *: Significant as P value < 0.05.
According to our results, VAS was significantly lower at all times measured in lidocaine group than placebo group.
In agreement with our study, Wu et al.[18], declared that the pain scores were significantly lower in the lidocaine group than the placebo group.
| Table 3. Interleukin-6, time of first sounds, flatus and defecation of the studied groups | |||
|---|---|---|---|
| Group A (n = 30) | Group B (n = 30) | P | |
| Interleukin-6 | |||
| Preoperative Interleukin-6 | 1 ± 0.7 | 1.2 ± 0.6 | 0.401 |
| 8 h postoperative IL6 | 23 ± 3 | 34 ± 5 | < 0.001* |
| 24 h postoperative IL6 | 14 ± 3 | 26 ± 8 | < 0.001* |
| Time of first sounds (h) | 3.3 ± 1. | 3.3 ± 1. | < 0.001* |
| Time of first flatus (h) | 5 ± 1 | 5 ± 1 | 0.003* |
| Time of first defecation (h) | 10.4 ± 1.6 | 10.4 ± 1.6 | 0.013* |
This aligned with Ibrahim et al.[19], observed that the perioperative IVL injection had significant effect in reduction of postoperative pain compared to the control group.
However, Sarakatsianou et al.[15], found that the postoperative pain scores were insignificantly different between the lidocaine group and the control group. The disparity in results could be attributed to differences in the study areas and patients’ characteristics.
In this study, the duration of anesthesia and time of 1st analgesia were significantly delayed in lidocaine group than placebo group.
This was aligned with Ibrahim et al.[19], who illustrated that the perioperative IVL injection had significantly elevated the main pain free period compared to the control group.
This agreed with Hassan et al.[16], who demonstrated that the time of 1st analgesia was significantly delayed in lidocaine group than control group.
In the present study, the interleukin-6 was insignificantly different at preoperative between both groups. Interleukin-6 was significantly lower at 8 h after incision and 24 h after surgery in lidocaine group than placebo group.
Lidocaine exerts potent anti-inflammatory effects by inhibiting leukocyte activation and adhesion, thereby reducing the release of pro-inflammatory cytokines such as IL-6[20]. By suppressing neutrophil priming and mitigating oxidative stress, it helps attenuate the inflammatory cascade triggered by surgical trauma[21].
In the same line, Wu et al.[22], reported that the interleukin-6 was significantly lower in the lidocaine group than the placebo group.
On the other hand, Oliveira et al.[23], showed that there was no statistically significant difference in IL-6 concentration between groups. This discrepancy in the findings might be attributed to the difference in the volumes of lidocaine administrated between their study and ours.
Our results showed that the time of sounds, time of flatus and the time of defecation were significantly lower in lidocaine group than placebo group.
This was consistent with Ibrahim et al.[19], demonstrated that the perioperative IVL injection had significantly decreased time of flatus compared to the control group.
In the same line, Song et al.[24], found that the time of flatus and the time of defecation were significantly lower in lidocaine group than control group.
On the other hand, Paterson et al.[25], found that perioperative administration of 2% IVL did not improve return of gut function at 72 h. The differences in findings may be attributed to variations in geographic location and the specific surgical procedures performed.
The limitations of the study included that the study was single centered with a relatively small sample size, the study did not compare the effect of intraoperative IVL in different volumes and concentrations on postoperative bowel recovery and inflammation in elective intestinal surgery and the study did not compare the effect of intraoperative IVL in different types of surgeries.
Conclusions
Intraoperative IVL infusion improved postoperative pain control, delayed the need for analgesia, and reduced inflammation, as shown by lower interleukin-6 levels. Additionally, it significantly accelerated bowel recovery, evidenced by earlier bowel sounds, flatus, and defecation, highlighting its potential to enhance postoperative recovery in elective intestinal surgery.
Referencias
1.Beaussier M, Delbos A, Maurice-Szamburski A, Ecoffey C, Mercadal L. Perioperative use of intravenous lidocaine. Drugs. 2018 Aug;78(12):1229-46. https://doi.org/10.1007/s40265-018-0955-x PMID:30117019
2.Yang W, Yan S, Yu F, Jiang C. Appropriate duration of perioperative intravenous administration of lidocaine to provide satisfactory analgesia for adult patients undergoing colorectal surgery: a meta-analysis of randomized controlled trials. LWW; 2023. pp. 494-506.
3.Eipe N. Intravenous Lidocaine. 1 ed: Springer, Cham; 2018. 83-103 p.
4.Baker ME, Hull T, Holubar S, Lightner A, Qazi T. Clinical issues facing pouch patients: an introduction to a special issue on the ileal pouch. Abdom Radiol (NY). 2023 Sep;48(9):2913-7. https://doi.org/10.1007/s00261-022-03723-x PMID:36334124
5.Paterson H, Vadiveloo T, Innes K, Balfour A, Atter M, Stoddart A, et al. Intravenous Lidocaine for Gut Function Recovery in Colonic Surgery. JAMA [Internet]. 2025 Jan 7;333(1):39. Available from: http://dx.doi.org/10.1001/jama.2024.23898 PMID:39602290
6.Bi Y, Diao M, Tao Y, Li H, Lin X. A Systematic Review and Meta-analysis of the Analgesic Effects of Lidocaine Administered Intravenously or Intraperitoneally Post-Abdominal Surgery. Pain Physician. 2024 Mar;27(3):E317-26. https://doi.org/10.36076/ppj.2024.27.E317 PMID:38506678
7.Weibel S, Jelting Y, Pace NL, Helf A, Eberhart LH, Hahnenkamp K, et al. Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery in adults. Cochrane Database Syst Rev. 2018 Jun;6(6):CD009642. https://doi.org/10.1002/14651858.CD009642.pub3 PMID:29864216
8.Lee IW, Schraag S. The use of intravenous lidocaine in perioperative medicine: anaesthetic, analgesic and immune-modulatory aspects. J Clin Med. 2022 Jun;11(12):3543. https://doi.org/10.3390/jcm11123543 PMID:35743617
9.Bezu L, Akçal Öksüz D, Bell M, Buggy D, Diaz-Cambronero O, Enlund M, et al.; The EuroPeriscope Group. Perioperative Immunosuppressive Factors during Cancer Surgery: An Updated Review. Cancers (Basel). 2024 Jun;16(13):2304. https://doi.org/10.3390/cancers16132304 PMID:39001366
10.Kuo CP, Jao SW, Chen KM, Wong CS, Yeh CC, Sheen MJ, et al. Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain and bowel function in patients undergoing colonic surgery. Br J Anaesth. 2006 Nov;97(5):640-6. https://doi.org/10.1093/bja/ael217 PMID:16952918
11.Courtney A, Clymo J, Dorudi Y, Moonesinghe SR, Dorudi S. Scoping review: the terminology used to describe major abdominal surgical procedures. World J Surg. 2024 Mar;48(3):574-84. https://doi.org/10.1002/wjs.12084 PMID:38342951
12.Jensen MS, Eriksen VR, Rasmussen SS, Meyhoff CS, Aasvang EK. Time to detection of serious adverse events by continuous vital sign monitoring versus clinical practice. Acta Anaesthesiol Scand. 2025 Jan;69(1):e14541. https://doi.org/10.1111/aas.14541 PMID:39468756
13.Pirie K, Traer E, Finniss D, Myles PS, Riedel B. Current approaches to acute postoperative pain management after major abdominal surgery: a narrative review and future directions. Br J Anaesth. 2022 Sep;129(3):378-93. https://doi.org/10.1016/j.bja.2022.05.029 PMID:35803751
14.Small C, Laycock H. Acute postoperative pain management. Br J Surg. 2020 Jan;107(2):e70-80. https://doi.org/10.1002/bjs.11477 PMID:31903595
15.Sarakatsianou C, Perivoliotis K, Baloyiannis I, Georgopoulou S, Tsiaka A, Tzovaras G. Efficacy of intraoperative intravenous lido¬caine infusion on postoperative opioid consumption after laparoscopic cholecystectomy: a randomized controlled trial. Langen- becks Arch Surg. 2023 May;408(1):197. https://doi.org/10.1007/s00423-023-02937-x PMID:37198418
16.Hassan MM, Saleh EG, Abdalla NO, Radwan NH, Abdelghfar EM. Effect of lidocaine infusion compared to dexmedetomidine infusion on proinflammatory cytokines and stress response in pelvi abdominal cancer surgeries: A randomized clinical trial. Anaesth Pain Intensive Care. 2022;26(1):44-52. https://doi.org/10.35975/apic.v26i1.1765
17.Choi SJ, Kim MH, Jeong HY, Lee JJ. Effect of intraoperative lidocaine on anesthetic consumption, and bowel function, pain intensity, analgesic consumption and hospital stay after breast surgery. Korean J Anesthesiol. 2012 May;62(5):429-34. https://doi.org/10.4097/kjae.2012.62.5.429 PMID:22679539
18.Wu Y, Chen Z, Yao C, Sun H, Li H, Du X, et al. Author Correction: Effect of systemic lidocaine on postoperative quality of recovery, the gastrointestinal function, inflammatory cytokines of lumbar spinal stenosis surgery: a randomized trial. Sci Rep. 2024 Apr;14(1):8480. https://doi.org/10.1038/s41598-024-59219-9 PMID:38605216
19.Ibrahim ASM, El-Shazly Ghanim A, Abdelaal WAM. Effect of intra operative intravenous lidocaine infusion on control of pain and recovery of intestinal function following surgical appendectomy. Qjm-Int J Med. 2023;116(1). https://doi.org/10.1093/qjmed/hcad069.008
20.Karnina R, Arif SK, Hatta M, Bukhari A. Molecular mechanisms of lidocaine. Ann Med Surg (Lond). 2021 Aug;69(2):102733. PMID:34457261
21.Jiang D, Muschhammer J, Qi Y, Kügler A, de Vries JC, Saffarzadeh M, et al. Suppression of neutrophil-mediated tissue damage a novel skill of mesenchymal stem cells. Stem Cells. 2016 Sep;34(9):2393-406. https://doi.org/10.1002/stem.2417 PMID:27299700
22.Wu Y, Chen Z, Yao C, Sun H, Li H, Du X, et al. Effect of systemic lidocaine on postoperative quality of recovery, the gastrointestinal function, inflammatory cytokines of lumbar spinal stenosis surgery: a randomized trial. Sci Rep. 2023 Oct;13(1):17661. https://doi.org/10.1038/s41598-023-45022-5 PMID:37848527
23.Oliveira CM, Sakata RK, Slullitel A, Salomäo R, Lanchote VL, Issy AM. [Effect of intraoperative intravenous lidocaine on pain and plasma interleukin-6 in patients undergoing hysterectomy]. Rev Bras Anestesiol. 2015;65(2):92-8. https://doi.org/10.1016/j.bjan.2013.07.017 PMID:25740274
24.Song X, Sun Y, Zhang X, Li T, Yang B. Effect of perioperative intravenous lidocaine infusion on postoperative recovery following laparoscopic Cholecystectomy-A randomized controlled trial. Int J Surg. 2017 Sep;45(2):8-13. https://doi.org/10.1016/j.ijsu.2017.07.042 PMID:28705592
25.Paterson H, Vadiveloo T, Innes K, Balfour A, Atter M, Stoddart A, et al. Intravenous lidocaine for gut function recovery in colonic surgery: A randomized clinical trial. JAMA. 2025 Jan;333(1):39- 48. https://doi.org/10.1001/jama.2024.23898 PMID:39602290

 ORCID
ORCID